Severe asthma and disease heterogeneity in chronic rhinosinusitis with nasal polyps
Severe asthma in CRSwNP
Authors
Abstract
Aim Chronic rhinosinusitis with nasal polyps (CRSwNP) is a heterogeneous inflammatory disease frequently associated with asthma, particularly severe asthma, which may influence disease phenotype and prognosis. However, real-world data on how severe asthma affects the clinical, inflammatory, and radiological features of CRSwNP remain limited.
Methods This single-center, retrospective, observational cohort study included adult patients diagnosed with CRSwNP between January 2020 and June 2024. Patients were classified according to the presence or absence of comorbid severe asthma, defined by Global Initiative for Asthma (GINA) criteria. Demographic characteristics, comorbidities, inflammatory biomarkers (peripheral blood eosinophil count [PBEC] and serum total IgE), symptom burden (VAS and SNOT-22), and radiological severity assessed by the Lund–Mackay (LM) score were compared between groups.
Results Among 52 patients included, 37 (71.2%) had comorbid severe asthma. Patients with severe asthma showed significantly higher PBEC (p = 0.045), while serum total IgE levels were comparable between groups. Obesity (65.7% vs 26.7%, p = 0.012) and nonsteroidal anti-inflammatory drug–exacerbated respiratory disease (NERD) (45.9% vs 6.7%, p = 0.006) were more prevalent in the asthma group. In contrast, symptom severity and radiological disease extent did not differ significantly.
Conclusion CRSwNP patients with comorbid severe asthma display a distinct phenotype marked by increased systemic eosinophilia and higher rates of NERD and obesity, despite similar symptom burden and radiological severity. These findings support integrating asthma severity and inflammatory biomarkers into multidimensional risk stratification and personalized management of CRSwNP.
Keywords
Introduction
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a heterogeneous, chronic inflammatory disease of the upper airway characterized by bilateral inflammatory nasal polyps originating from the mucosa of the nasal cavity and paranasal sinuses.1 According to the European Position Paper on Rhinosinusitis and Nasal Polyps 2023 (EPOS 2023), CRSwNP is defined by the presence of at least two sinonasal symptoms persisting for a minimum of 12 weeks, with nasal obstruction/congestion or nasal discharge being mandatory, accompanied by facial pain/pressure and/or a reduction or loss of the sense of smell, together with the detection of bilateral nasal polyps on nasal endoscopy and/or characteristic changes on computed tomography (CT) imaging.2
With an estimated prevalence of approximately 1–4% in the general population, CRSwNP represents a substantial clinical and socioeconomic burden because of its chronic course, tendency to recur, and frequent refractoriness to standard therapies.1,2 Despite guideline-directed intranasal corticosteroid therapy and endoscopic sinus surgery (ESS), a considerable proportion of patients experience persistent symptoms, recurrent polyp formation, and the need for multiple surgical interventions.2,3 This recurrent disease course often necessitates repeated use of systemic corticosteroids and leads over time to increased morbidity related to cumulative corticosteroid exposure and associated adverse effects.4,5 These features underscore the heterogeneity of CRSwNP and explain why responses to standard treatments vary markedly among patients.
Asthma is one of the most common and clinically relevant comorbidities in CRSwNP and represents a prototypical example of the “united airway disease” concept.6 This concept assumes that inflammation of the upper and lower airways is driven by shared pathogenetic mechanisms and constitutes different clinical manifestations of a single disease spectrum. Epidemiological data indicate that the prevalence of asthma among patients with CRSwNP may reach 45–70%, whereas CRSwNP is observed more frequently in patients with asthma, particularly adult-onset and severe asthma, than in the general population.6,7
In patients in whom asthma coexists with CRSwNP, sinonasal symptoms are more severe, health-related quality of life is more markedly impaired, systemic corticosteroid requirements are higher, and the likelihood of undergoing surgical intervention is increased.6 Conversely, the presence of CRSwNP is associated with poorer asthma control, a higher frequency of exacerbations, and a more pronounced decline in lung function.8
Nonsteroidal anti-inflammatory drug–exacerbated respiratory disease (NERD) represents one of the most severe and prototypical forms of united airway disease, characterized by the coexistence of CRSwNP, asthma, and intolerance to nonsteroidal anti-inflammatory drugs (NSAIDs).9 In this patient group, nasal polyps typically follow a more aggressive course, recur early after surgery, and exhibit marked treatment resistance. The presence of NERD is therefore recognized as a strong risk factor for severe disease and poor prognosis in CRSwNP.8,9
One of the principal mechanisms linking CRSwNP and asthma is a shared type 2 (T2) inflammatory endotype.10 In Western countries and in many Asian populations, CRSwNP is predominantly characterized by T2 inflammation, marked by eosinophilic infiltration, increased total and local immunoglobulin E (IgE) levels, and overexpression of cytokines such as interleukin-4, -5, and -13.11 T2-high inflammation is associated with a more severe disease course, an increased risk of postoperative recurrence, greater systemic corticosteroid requirements, and a poorer response to conventional therapies.11 This inflammatory profile appears to be particularly pronounced in patients with CRSwNP and severe asthma, who constitute the most difficult-to-treat patient subgroup.11,12
In current clinical practice, disease severity and treatment outcomes in CRSwNP are increasingly assessed using multidimensional measures that integrate structural, symptomatic, inflammatory, and treatment-related parameters. The Lund–Mackay (LM) score derived from CT provides an objective measure of sinus involvement and is associated with disease extent and the likelihood of requiring surgery.2,13 Symptom burden and health-related quality of life are commonly assessed using the 22-item Sino-Nasal Outcome Test (SNOT-22), which is widely used as a core patient-reported outcome measure in CRSwNP studies.2,14
Inflammatory biomarkers, such as peripheral blood eosinophil counts (PBEC) and serum total IgE levels, are gaining increasing importance in defining disease phenotypes, predicting prognosis, and identifying candidates for targeted biologic therapies.10 In addition, a history and number of prior polypectomies are considered pragmatic indicators of long-term recurrence risk and treatment refractoriness.4
Although the coexistence of CRSwNP and asthma has been extensively described in the literature, real-world data remain limited regarding the extent to which the presence of
severe asthma (SA) modifies the clinical, laboratory, and radiological characteristics of CRSwNP.6,10 A clearer understanding of how asthma severity influences sinonasal disease burden, inflammatory biomarkers, surgical recurrence, and treatment resistance is essential to improve patient risk stratification and to develop more individualized treatment strategies.9
This study classified patients into two main groups based on the presence or absence of asthma. All patients with asthma were further categorized as having SA according to the Global Initiative for Asthma (GINA) criteria.15 Clinical characteristics, laboratory findings, and paranasal sinus computed tomography (PNS CT) parameters were compared between these groups.
Materials and Methods
Study DesignThis study was designed as a single-center, retrospective, observational cohort study including patients diagnosed with CRSwNP between January 2020 and June 2024. Patients were categorized into two groups based on the presence or absence of asthma; all patients with asthma were defined as having SA according to the GINA guidelines.15 Clinical, laboratory, and radiological characteristics were compared between the two groups.
Patients PopulationPatients aged 18 years and older who were diagnosed with CRSwNP according to the criteria of EPOS 2023 were included in the study.2 Based on comprehensive clinical and laboratory evaluations, all patients were classified into two main groups according to the GINA criteria: those with comorbid SA and those without asthma. GINA 2025, SA was defined as asthma that requires treatment with high-dose inhaled corticosteroids in combination with a second controller and/or systemic corticosteroids to prevent it from becoming uncontrolled, or that remains uncontrolled despite this optimized therapy.15
Patients with concomitant conditions that could influence type 2 inflammation and alter disease phenotype—such as hypereosinophilic syndrome, eosinophilic granulomatosis with polyangiitis (EGPA), and allergic bronchopulmonary aspergillosis (ABPA)—were excluded from the study.
Clinical, Laboratory, and Radiological AssessmentPatients’ demographic characteristics -age, gender, and body mass index (BMI)- and accompanying comorbidities (NERD and obesity), as well as serum total IgE levels and PBEC, were retrospectively recorded from the hospital database.
In addition, as part of the clinical evaluation performed at the initial outpatient visit, symptom severity was assessed using the Visual Analog Scale (VAS) and the SNOT-22, and PNS CT images were analyzed. LM scoring was performed based on PNS CT findings according to Supplementary Table 1.13 The VAS was evaluated such that a score of 0 represented the absence of symptoms, whereas a score of 10 indicated very severe symptoms.16
The SNOT-22 was administered using a validated 22-item patient-reported outcome measure that assesses sinonasal symptoms and health-related quality of life over the preceding two weeks, with each item scored on a scale from 0 to 5 (Supplementary Table 2).14
Ethical ApprovalThis study was approved by the Ethics Committee of Süreyyapaşa Chest Diseases and Thoracic Surgery Training and Research Hospital (Date: 2024-10-31, No: 2024-28).
Statistical AnalysisStatistical analyses were performed using the trial version of SPSS software version 22.0 (SPSS Inc., Chicago, IL, USA). The normality of distribution for quantitative variables was assessed using the Kolmogorov–Smirnov and Shapiro–Wilk tests. Comparisons between independent groups were conducted using the independent samples t-test for normally distributed variables and the Mann–Whitney U test for non-normally distributed variables. Associations between categorical variables were evaluated using the chi-square test. Descriptive statistics for normally distributed quantitative variables were presented as mean ± standard deviation, whereas non-normally distributed quantitative variables were expressed as median (minimum–maximum). Categorical variables were summarized as frequencies and percentages. A p-value < 0.05 was considered statistically significant.
Reporting GuidelinesThe study was reported in accordance with STROBE guideline.
Results
A total of 52 patients diagnosed with CRSwNP were included in the study. The mean age of the patients was 47.76 ± 11.03 years, and 24 (46.2%) were female. The mean BMI was 37.05 ± 14.02 kg/m². Comorbid SA was present in 37 patients (71.2%), whereas 15 patients (28.8%) had no asthma. In the overall cohort, the prevalence of atopy was 50%, and the prevalence of NERD was 34.6%. The median PBEC was 470 cells/µL (range, 20–6370), and the median serum total IgE level was 362 IU/mL (range, 23–3438). Obesity was present in 51.9% of the patients, and 67.3% had a history of prior polypectomy, with a mean number of polypectomies of 2.86 ± 2.04. The mean SNOT-22 score was 67.28 ± 19.32. On PNS CT, the mean total LM score was 14.43 ± 6.92 (Supplementary Table 3).
Comparison of Patients With Severe Asthma and Without AsthmaThe mean age of CRSwNP patients with comorbid SA was 49.35 ± 10.45 years, whereas it was 43.86 ± 11.83 years in those without asthma; no statistically significant difference in age was observed between the groups (p: 0.105). Although the proportion of female patients was higher in the SA group, this difference did not reach statistical significance (54.1% vs 26.7%; p: 0.067).
The prevalence of obesity was significantly higher in patients with SA (65.7%) compared with those without asthma (26.7%) (p = 0.012). Similarly, NERD was detected in 45.9% of patients with SA, whereas the prevalence was only 6.7% in the non-asthma group (p = 0.006). There was no significant difference between the groups with respect to atopy (p = 0.536).
The PBEC was significantly higher in patients with SA (620 [20–6370] vs 370 [100–1380] cells/µL; p = 0.045). In contrast, serum total IgE levels were comparable between the two groups (p: 0.840).
With regard to symptom severity, no statistically significant differences were observed between the groups in VAS scores for nasal congestion, loss of smell, or facial pressure/pain, nor in SNOT-22 scores (all comparisons p > 0.05). The mean total LM scores were also similar between patients with and without asthma (14.76 ± 7.48 vs 13.76 ± 5.87; p: 0.677), and no significant differences were found in sinus-specific subscores (Table 1). The detailed data are provided in Supplementary Tables 3-5.
Discussion
In this single-center real-world cohort, we evaluated the impact of comorbid SA on the clinical, inflammatory, and radiological characteristics of patients with CRSwNP. The demographic and disease-related features of our cohort were largely consistent with those reported in large surgical series, real-world registries, and biologic clinical trial populations, supporting the external validity of our findings.1,6,7,8,9,10 In particular, the close resemblance of baseline disease burden and patient profiles to those described in large-scale real-world registries suggests that our study population is representative of a tertiary-care, biologic-eligible CRSwNP cohort.17
The high prevalence of comorbid SA in our cohort (71.2%) lies at the upper end of reported ranges and indicates enrichment for more severe, type 2–high disease. This observation is in line with data from tertiary referral centers and biologic clinical trial screening populations.3,8,9,17 Similarly, the observed prevalence of NERD (34.6%) supports the concept that asthma–CRSwNP comorbidity may be associated with a more refractory “united airways” phenotype.18
From an inflammatory perspective, the significantly higher PBEC observed in patients with comorbid SA, in contrast to comparable serum total IgE levels between groups, is noteworthy. This finding is consistent with real-world studies suggesting that eosinophilia may represent a more sensitive marker of type 2 inflammatory burden in asthma-associated CRSwNP than total IgE alone.5,12,14,19 The absence of a parallel increase in total IgE further underscores the heterogeneous nature of type 2 inflammation and highlights the limitations of relying on a single biomarker to fully characterize disease biology.
In addition, the significantly higher prevalences of NERD and obesity in the severe asthma group reveal a clinically relevant clustering of comorbidities. The coexistence of asthma, eosinophilia, NERD, and metabolic comorbidities is consistent with the concept of a more severe, type 2–high, and treatment-refractory united airway phenotype.4,14,16,17,18 Recent studies have also emphasized the potential role of obesity in modulating disease severity and response to biologic therapies in CRSwNP, further enhancing the clinical relevance of our findings.17,18,19
Despite these inflammatory and comorbidity-related differences, baseline symptom burden and radiologic disease extent did not differ significantly between patients with and without asthma. Comparable SNOT-22 scores, VAS symptom assessments, and LM scores across groups are in agreement with findings from large real-world registries and observational studies.3,10,11,15,17 These observations suggest that cross-sectional symptom- and CT-based measures may not adequately reflect biological aggressiveness or long-term risk in asthma-associated CRSwNP.
In this context, longitudinal data from the literature provide important insights. Previous studies have demonstrated that the presence of asthma increases postoperative recurrence risk and amplifies the prognostic impact of eosinophilia in CRSwNP.6,13,15 More recent analyses and predictive modeling studies have further identified asthma as an independent risk factor for disease recurrence and persistence.17,18 Although long-term outcomes were not assessed in the present study, the enrichment of eosinophilia, NERD, and obesity within the SA subgroup supports the hypothesis that these patients may follow a more aggressive and recurrence-prone disease course over time.
From a clinical perspective, our findings indicate that risk assessment in CRSwNP should not be limited to traditional severity metrics such as SNOT-22 and LM scores. Incorporating asthma severity, systemic inflammatory biomarkers, and key comorbidities into a multidimensional evaluation framework may improve risk stratification and facilitate earlier and more individualized transitions to targeted biologic therapies. 3,8,10,17,18,19
Limitations
This study has several limitations. Its retrospective, single-center design may limit generalizability and introduce selection bias. In addition, the relatively small sample size—particularly in the non-asthma group—and the lack of longitudinal follow-up data preclude direct assessment of postoperative recurrence and long-term treatment outcomes.
Conclusion
Patients with CRSwNP and comorbid severe asthma exhibit a distinct inflammatory and comorbidity profile characterized by higher peripheral eosinophil counts and increased prevalence of NERD and obesity, despite comparable symptom burden and radiological severity. These findings underscore the importance of incorporating asthma severity and systemic inflammatory markers into multidimensional risk stratification and personalized management strategies for CRSwNP.
Declarations
Ethics Declarations
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study protocol was reviewed and approved by the relevant institutional ethics committee.
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent was waived by the Ethics Committee due to the retrospective design of the study and the use of anonymized patient data.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Author Contributions (CRediT Taxonomy)
Conceptualization: Ş.Ö., F.M.T.
Methodology: Ş.Ö., F.M.T.
Study Design: Ş.Ö., F.M.T.
Data Collection: Ş.Ö., F.M.T.
Data Curation: Ş.Ö, F.M.T.
Formal Analysis: Ş.Ö., F.M.T.
Investigation: Ş.Ö., F.M.T.
Resources: Ş.Ö., F.M.T.
Writing – Original Draft Preparation: Ş.Ö., F.M.T.
Writing – Review & Editing: Ş.Ö, F.M.T
Visualization: Ş.Ö., F.M.T.
Supervision: Ş.Ö., F.M.T.
Project Administration: Ş.Ö., F.M.T.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
Abbreviations
ABPA: Allergic bronchopulmonary aspergillosis
BMI: Body mass index
CRSwNP: Chronic rhinosinusitis with nasal polyps
CT: Computed tomography
EGPA: Eosinophilic granulomatosis with polyangiitis
EPOS: European Position Paper on Rhinosinusitis and Nasal Polyps
ESS: Endoscopic sinus surgery
GINA: Global Initiative for Asthma
IgE: Immunoglobulin E
LM: Lund–Mackay
NERD: Nonsteroidal anti-inflammatory drug–exacerbated respiratory disease
NSAIDs: Nonsteroidal anti-inflammatory drugs
PBEC: Peripheral blood eosinophil count
PNS CT: Paranasal sinus computed tomography
SA: Severe asthma
SNOT-22: 22-item Sino-Nasal Outcome Test
SPSS: Statistical Package for the Social Sciences
T2: Type 2
VAS: Visual Analog Scale
References
-
Chen CC, Buchheit KM. Endotyping chronic rhinosinusitis with nasal polyps: understanding inflammation beyond phenotypes. Am J Rhinol Allergy. 2023;37(2):132-139. doi:10.1177/19458924221149003
-
Fokkens WJ, Viskens AS, Backer V, et al. EPOS/EUFOREA update on indication and evaluation of biologics in chronic rhinosinusitis with nasal polyps 2023. Rhinology. 2023;61(3):194-202. doi:10.4193/Rhin22.489
-
Chen YS, Feng CY, Su SH, et al. Recurrence of chronic rhinosinusitis with nasal polyps after surgery: risk factors, predictive models, and treatment approaches with a focus on western and Asian differences. Medicina (Kaunas). 2025;61(9):1620. doi:10.3390/medicina61091620
-
Peters AT, Pinto JM, Buchheit KM, et al.The burden of systemic corticosteroids in patients with chronic rhinosinusitis with nasal polyps. Allergy Asthma Proc. 2025;46(5):406-413. doi:10.2500/aap.2025.46.250050
-
Davis GE, Zeiger RS, Emmanuel B, et al. Systemic corticosteroid-related adverse outcomes and health care resource utilization and costs among patients with chronic rhinosinusitis with nasal polyposis. Clin Ther. 2022;44(9):1187-1202. doi:10.1016/j.clinthera.2022.08.004
-
Laidlaw TM, Mullol J, Woessner KM, Amin N, Mannent LP. Chronic rhinosinusitis with nasal polyps and asthma. J Allergy Clin Immunol Pract. 2021;9(3):1133-1141. doi:10.1016/j.jaip.2020.09.063
-
Promsopa C, Kansara S, Citardi MJ, Fakhri S, Porter P, Luong A. Prevalence of confirmed asthma varies in chronic rhinosinusitis subtypes. Int Forum Allergy Rhinol. 2016;6(4):373-377. doi:10.1002/alr.21674
-
Castillo JA, Plaza V, Rodrigo G, et al. Chronic rhinosinusitis with nasal polyps and allergic rhinitis as different multimorbid treatable traits in asthma. J Allergy Clin Immunol Glob. 2023;2(4):100134. doi:10.1016/j.jacig.2023.100134
-
Walters BK, Hagan JB, Divekar RD, et al. Aspirin-exacerbated respiratory disease and the unified airway: a contemporary review. Otolaryngol Clin North Am. 2023;56(1):107-124. doi:10.1016/j.otc.2022.09.008
-
Bachert C, Zhang N, Holtappels G, et al. Presence of IL-5 protein and IgE antibodies to staphylococcal enterotoxins in nasal polyps is associated with comorbid asthma. J Allergy Clin Immunol. 2010;126(5):962-968, 968.e1-e6. doi:10.1016/j.jaci.2010.07.007
-
Wang X, Zhang N, Bo M, et al. Diversity of THcytokine profiles in patients with chronic rhinosinusitis: a multicenter study in Europe, Asia, and Oceania. J Allergy Clin Immunol. 2016;138(5):1344-1353. doi:10.1016/j.jaci.2016.05.041
-
Tomassen P, Vandeplas G, Van Zele T,et al. Inflammatory endotypes of chronic rhinosinusitis based on cluster analysis of biomarkers. J Allergy Clin Immunol. 2016;137(5):1449-1456.e4. doi:10.1016/j.jaci.2015.12.1324
-
Okushi T, Nakayama T, Morimoto S,et al. A modified Lund-Mackay system for radiological evaluation of chronic rhinosinusitis. Auris Nasus Larynx. 2013;40(6):548-553. doi:10.1016/j.anl.2013.04.010
-
Hopkins C, Gillett S, Slack R, Lund VJ, Browne JP. Psychometric validity of the 22-item Sinonasal Outcome Test. Clin Otolaryngol. 2009;34(5):447-454. doi:10.1111/j.1749-4486.2009.01995.x
-
Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention 2025. Accessed January 19, 2026. https://ginasthma.org
-
Ciprandi G, La Mantia I. VAS for assessing the perception of antihistamines use in allergic rhinitis. Acta Biomed. 2019;90(7-S):41-44. doi:10.23750/abm.v90i7-S.8657
-
Desrosiers M, Laidlaw TM, White AA, et al. Prevalence of nonsteroidal anti-ınflammatory drug-exacerbated respiratory disease among patients with chronic rhinosinusitis with nasal polyps in the global AROMA registry. Int Arch Allergy Immunol. 2025;25:1-6. doi:10.1159/000548186
-
Buchheit KM, Vandewalle E, Elzinga HBE,et al. Efficacy of biologics in NSAID-ERD: united airways from the nose to the bronchi. J Allergy Clin Immunol Pract. 2024;12(11):2917-2932. doi:10.1016/j.jaip.2024.09.021
-
Peters AT, Heffler E, Buchheit KM,et al.Comparing baseline characteristics of patients with chronic rhinosinusitis with nasal polyps with and without asthma in the AROMA registry. J Asthma Allergy. 2025;18(6):1041-1049. doi:10.2147/JAA.S519901
Tables
Table 1. Comparison of demographic, clinical, and inflammatory characteristics of patients with chronic rhinosinusitis with nasal polyps according to asthma status
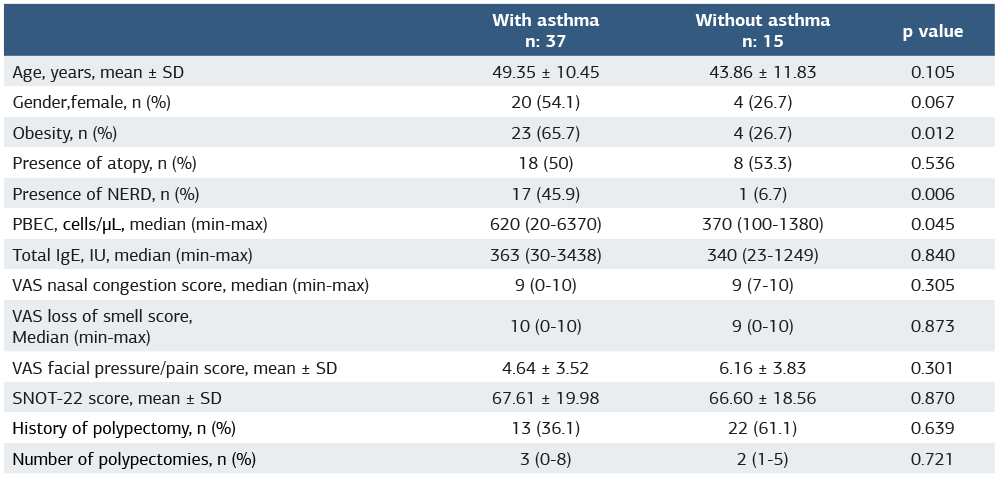
Abbreviations: SD = Standart Deviation; NERD = Non-steroidal anti-inflammatory drug–exacerbated respiratory disease; PBEC = Peripheral Blood Eosinophil Count ; Ig= Immunoglobulin; VAS = Visual Analog Scale ; SNOT-22 = Sinonasal Outcome Test-22.
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Şeyma Özden, Fatma Merve Tepetam. Severe asthma and disease heterogeneity in chronic rhinosinusitis with nasal polyps. Ann Clin Anal Med 2026;17(Suppl 2):S157-161. doi: 10.4328/ACAM.50053
- Received:
- January 25, 2026
- Accepted:
- March 9, 2026
- Published Online:
- March 11, 2026
- Printed:
- March 20, 2026

