In vitro assessment of wear in peripheral nerve block needles for re-use
Wear in nerve block needles
- Received:
- January 15, 2025
- Accepted:
- March 3, 2025
- Published Online:
- March 13, 2025
- Printed:
- October 1, 2025
Authors
Abstract
Aim This study aims to assess the safety and effectiveness of reusing peripheral nerve block needles, focusing on electrical resistance and immersion forces. Peripheral nerve block needles are typically single-use, but reusing them could reduce costs associated with needle disposal and purchase. While safety concerns exist, especially regarding infection and needle blunting, these issues need to be thoroughly evaluated.
Methods We measured immersion loads and electrical resistances of 20 unused peripheral nerve block needles. Four anesthetists applied loads to a model tissue with these needles. Nine needles were selected for reuse, assigned to anesthetists, and immersed in model tissue at 2.5 cm depth. After rinsing with isotonic solution, the needles underwent sterilization, and this cycle was repeated 20 times.
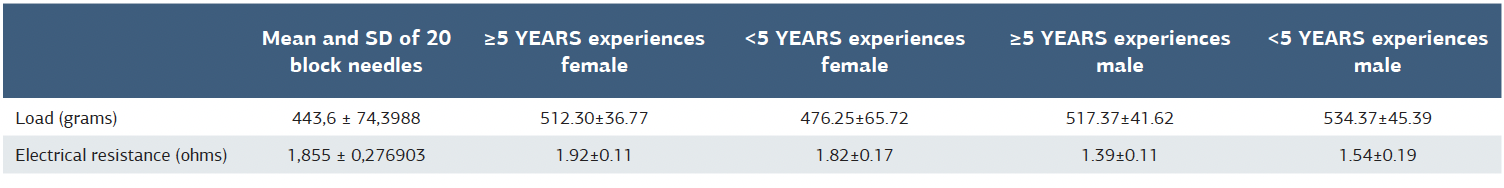
Results The mean load and electrical resistance of the initial 20 needles were 443.6 ± 74.39 and 1.855 ± 0.27, respectively. No significant differences in electrical resistance or load measurements were noted among anesthetists with varying experience levels (p-values ≥ 0.150). Likewise, no statistical difference existed between the initial and reused needles (p = 0.265).
Conclusion The study indicates no significant difference in forces applied by male and female practitioners or across experience levels, suggesting that the reuse of peripheral nerve block needles is viable. The needles can safely undergo at least 20 cycles of reuse using the employed sterilization technique without compromising electrical conduction or immersion efficacy.
Keywords
Introduction
Medical products, including peripheral nerve block needles, are mainly manufactured for single use. Although peripheral nerve block needles are cheaper, they are used in much more numbers. Reusing peripheral nerve block needles was considered a severe saving tool 1,2,3. Both the needle price and the need to dispose of used needles as medical waste increases the cost. (Needle unit price is about 12 $-Invoiced to the institution, Sterilization costs 0.2 $ per needle (The Institutional conditions)). Also, this approach is compatible with producing less waste (less CO2 emission) 4. The needles have been reported to be effectively sterilized by current techniques 2,3. The sterilization cost is meager compared to the needle unit price.
For this reason, the objective evaluation of the wear problem caused by reuse is essential. If they are reused, the reuse procedure should be proofed that it’s safe, and there must be an identified(and also written) reuse procedure. It is envisaged that physical wear of the peripheral block needles will result in breakage of the needles, loss of transmitting cables, and reduced transmission of electricity. The peripheral nerve block needles work with the principle of getting a contraction response from the relevant muscle tissue by giving low-voltage electrical current to the nerves. Healthy electrical conduction means the robust operation of a nerve stimulator.
On the other hand, our study includes proven sterilization methods for peripheral nerve block needles, and our results are thought to improve reuse standards. The cycle determined by our study can be used alone as a standard of reuse. The study is planned on these principles.
According to our hypothesis, each time the needle is used (every time it is immersed in the artificial tissue), it should be a little blunter, it should be pushed into the tissue with a bit more force the next time, and also the electrical resistance should increase with wear. If these increases do not occur, it will be seen that the functionality of the needles does not decrease in reuse. In addition, it has been aimed to determine whether chemicals, temperature, and other unpredictable factors used for sterilization have a corrosive effect on the needle and whether these effects affect the needle’s operability. This entire cycle will be expressed with numerical data and analyzed by statistical methods. It has been investigated whether there is a significant difference between the interventions in applied load and resistance to electric current.
Materials and Methods
Firstly, to numerically express the electrical resistance of the peripheral nerve block needles and the immersion forces in the model tissue, the immersion loads and average electrical resistances of the 20 unused needles were measured.
The male participant, who had 5 years or more experience, was chosen as the practitioner in the random selection between the 4 participants anesthetists to apply the load of the first 20 needles to the model tissue. In our study, the Pajunk® Sonoplex Stim cannula model 22 gauge 50 mm peripheral nerve block needle was used. As model tissue, Genesis® brand, Spinal Injection Smilation (model code 800.866.3342) has been used. Model artificial working tissue was placed on sensitive scales (Swan® Digital Precision (2018 China) scales (with an accuracy of ± 0.1 g)) and the block needle was immersed to a depth of 2,5 cm, the highest values (force in-grams applied to the needle) recorded with the measuring instrument were recorded and the average values were calculated. In the next stage, the electrical resistance of each needle (T-Technic A830L (2017 China) digital multimeter) was measured with a multimeter (recorded in ohms). The calculation of these values has been determined as a reference of the mean for the electrical resistance and force required for immersion of an average needle. That data would able to be used for determining and comparison with the increase/decrease in the force requirement and electrical resistance changes in successive interventions with a single needle in subsequent measurements.
In the second stage, 9 of the needles were randomly selected and divided into five groups. Two needles were given to each anesthetist. One needle was reserved as a reference for comparison of visible physical wear.
The needles were immersed in the model working tissue, at a depth of 2.5 cm, perpendicular to the ground (the force applied to the needle was detected by tare), and after the measurements were taken, the needles were rinsed with an isotonic solution. They were then sent to the sterilization unit. The needles packaged in the sterilization unit were sterilized in a CASP brand CASP-120 model sterilization device with 55-60 % hydrogen peroxide at 45±5 °C in 53 minutes, and a cycle was completed. This described cycle was repeated 20 times, and data was recorded. The force applied to the needle was recorded in grams (gr), and the measured electrical resistance was in ohms (Ω). Thus, 20 cycles of use, mechanical fatigue, and chemical contact cycles were simulated. The metal alloy of the needle used was worn fairly by exposure to mechanical trauma and chemicals. This whole cycle will be expressed with numerical data and analyzed by statistical methods, it is investigated whether there is a significant difference in terms of the power applied between the enterprises. The measuring device is shown in Figures 1, 2, and 3. The study was carried out at an ambient temperature of 18-22 °C to avoid temperature effects. The cycle described above was performed by a total of 4 anesthetists. Visible signs of wear (bending, twisting, disintegration, etc.) occurring in the needles were also added to the data, if any, were noted.
Statistical Analysis
When histogram plot analysis was performed, the distribution of measurements was parametric. When the Skewness and Kurtosis normality analysis and confidence interval values were examined, Repeated Measures ANOVA tests for proportions were used to assess the correct identification of each of the four anesthesiologists. The 0.05 level of significance was used to reject the null hypothesis. Power analysis showed that a sample of 20 times of use will yield 80 % power to detect a difference in the proportion of correct identifications of each four anesthesiologists. Statistical analyses were performed using IBM SPSS Statistics version 22 (IBM) and Rex version
3.5.3 (RexSoft Inc).
Ethical Approval
This study was approved by the Ethics Committee of the Institutional Review Board of Alanya Alaaddin Keykubat University Medicine Faculty (Date: 2022-05-13, No:10354421).
Results
The mean load and electrical resistance measurements of the first 20 block needles are 443.6 ± 74.39 and 1.855 ± 0.27 with their standard deviations(Table 1). A total of four anesthetists were included in the study (n = 4). When histogram plot analysis was performed, the distribution of measurements was non- parametric. When the Skewness and Kutosis normality analysis and confidence interval values were examined, no statistical significance was found in the measurements. In this sense, p-value was found to be 0.163 and 0.308, respectively, in load measurements, those with 5 years or more and less than 5 years (Coinfidence Interval = -0.719 / -0.064). For electrical resistance measurements, p values were found to be 0.150 and 0.361 for those with 5 years and more experience and less than 5 years (Confidence Interval = 0.540 / 1.110). In the analyses made with the Repeatedy ANOVA test, no statistically significant difference was observed in the load and electrical resistance measurement values in those with 5 years and more experience and less than 5 years (p-value = 0.587 and 0.250 respectively) (Table 2). The mean load measurements (505.31 ± 63.87) in participants with less than 5 years of experience were less than those with 5 years or more (514 ± 39.95). In addition, whether the experience is an effective factor was evaluated with the Logistic Regression test. Accordingly, there was not a sufficient statistical difference between the groups (Adjusted R square = - 0.004, p < 0.498). The average of electrical resistance measurements (1.68 ± 0.30) in the participants with less than 5 years of experience was higher than those with 5 years of experience (1.65 ± 0.37). In addition, whether the experience is an effective factor was evaluated with the Logistic Regression test. Accordingly, there was not a sufficient statistical difference between the groups (Adjusted R square = - 0.005, p <0.597). There was no statistical difference between the mean load the first 20 block needle and the mean loads of participants. (p = 0,265) Also, there was no statistical difference between the mean electrical resistance of the first 20 block needle and the mean loads of participants. (p = 0.117)(Table 3).
Discussion
The medical equipment reuse debate has been ongoing for a long time. There are discussions on many issues, such as principles to be followed, procedures to be applied, and the selection of sterilization/disinfection methods. The reason for the reuse of medical equipment may be economical 1, environmental 2, or different reasons related to access to medical equipment. It has been reported that they can be used safely by adhering to the principles regarding reuse 2,3. However, written procedures based on clinical studies should be applied, a person responsible for daily operations (reuse manager) should be determined, use should be found on the patient’s knowledge, and criteria should be developed to determine how much time a device/material can be used before wearing away. Also, ‘wearing’ should be described for each medical device/equipment 1. The things to be done for the first three criteria are clear, but for the fourth criterion, it is necessary to work separately for each device/ material as stated.
The debate regarding the reuse of medical equipment is multi-faceted, enveloping a wide range of issues such as the principles to be adhered to, applicable procedures, and the selection of suitable sterilization or disinfection methods. The rationale for reusing medical equipment is multi-dimensional, involving economic incentives, environmental considerations, and access-related challenges. While economic motivations signify cost reductions 1, environmental concerns underline the need for sustainable practices 3,4,5, and access issues highlight the necessity to make medical equipment available in resource-limited settings.
Historically, the reuse of medical devices has been met with skepticism due to concerns about safety and efficacy. However, numerous studies suggest that these devices can be safely reused if stringent protocols are followed. Key to this process is the implementation of standardized procedures grounded in clinical research, designating a responsible individual or ‘reuse manager’ to oversee daily operations. Furthermore, patients should be informed about potential reuse, and specific criteria must be established to assess the lifespan and wear of each device or material. The concept of ‘wear’ must be clearly defined for each piece of medical equipment to ensure patient safety 3,4,6.
In our comprehensive study, we explored the reuse potential of nerve block needles utilized in regional anesthesia. These needles were subjected to a rigorous sterilization process, proven for their efficacy, and reused up to 20 times. Our findings demonstrated that the needles retained sufficient strength and sharpness, with no notable change in the force required to pierce tissue. Moreover, the sterilization process did not alter the electrical resistance of the needles, ensuring their ability to safely conduct electrical stimulation even after multiple uses. This aspect is crucial, as it assures that the needles maintain their functionality and reliability throughout their reuse cycle. The reuse of medical equipment extends beyond nerve block needles. For instance, the reuse of PPMs has shown significant economic advantages, providing life-saving devices to patients who lack the financial means to afford new units 1. Although nerve block needles are less costly compared to PPMs, the frequency of their use suggests that the financial gains from reuse could be substantial. In the clinical setting, the cost per nerve block needle is approximately $12, whereas the cost for sterilization and packaging for reuse is a mere $0.20. Consequently, this presents a potential savings of more than
$8.50 per needle. Additionally, the disposal costs for used block needles as medical waste are reduced, further contributing to economic savings.
From an environmental standpoint, the reduction of medical waste through the reuse of equipment is a critical factor. Decreasing the volume of medical waste not only cuts down disposal costs but also aids in reducing carbon dioxide emissions, thus contributing to environmental sustainability 3,7. The commitment to reducing the carbon footprint aligns with broader global initiatives aimed at combating climate change and promoting eco-friendly practices in healthcare.
Given the environmental benefits, healthcare facilities are increasingly considering the implementation of reuse policies. However, this requires careful planning and adherence to safety protocols to mitigate the risk of infection and ensure patient safety. The effectiveness of these protocols is pivotal, as it determines the feasibility and acceptance of medical device reuse on a wider scale.
Ultrasound (US) probes provide a compelling example of how reusable medical equipment can be integrated into clinical practice. In US-guided procedures, these probes can act as vectors for pathogen transmission, necessitating stringent disinfection protocols. The importance of US probe disinfection parallels that of other medical materials used in invasive procedures guided by ultrasound 5,6,8. While there have been few reported cases of infections linked to US probe disinfection techniques 7,8,9, these instances underline the necessity for effective sterilization methods that ensure patient safety without compromising the functionality of the devices.
In regional anesthesia practices that utilize ultrasound guidance, various disinfection methods are employed, each validated for their efficacy in different studies 10,11. The success of these methods indicates that with the proper protocols in place, medical devices such as US probes can be reused safely and effectively—a conclusion that can be extended to other medical devices under appropriate conditions.
In clinics that permit equipment reuse, institutions must adopt a comprehensive framework of principles to ensure safe reuse practices. This includes a detailed examination of potential problems associated with each specific type of reused device. Our study specifically investigated peripheral nerve block needles and found no evidence of needle tip blunting or compromised electrical conduction following multiple use cycles. Importantly, no visible damage was observed on these needle tips even when examined under 10x magnification (Figure 3). This finding is crucial, as it demonstrates the structural integrity and operational safety of the needles upon reuse.
Furthermore, variations in the electrical conduction of block needles present a potential concern in reuse practices. However, our study has provided a foundation for understanding these variations, contributing valuable insights to the literature where such assessments have been limited. The consistency in electrical conduction observed in our research underscores the notion that proper sterilization and handling can maintain the functional integrity of these devices.
Each medical device intended for reuse must have a defined “final number of use cycles” based on empirical evidence. For nerve block needles, our research suggests that up to 20 reuse cycles are both safe and cost-effective. This level of reuse can reduce expenditures for each needle by more than 80%, highlighting significant cost efficiencies without sacrificing safety or performance.
Despite the apparent benefits, the reuse of medical devices is not without challenges. One primary concern is the potential for infection or cross-contamination if sterilization is not meticulously performed. This underscores the necessity for rigorous training and adherence to established protocols by medical personnel tasked with sterilizing and reusing equipment. Institutions must remain vigilant in monitoring compliance with these standards to safeguard against potential risks.
Moreover, patient perceptions and acceptance of reused medical devices can vary, influencing the broader adoption of reuse practices. Transparent communication with patients about the safety measures in place and the quality assurance processes followed is essential. Education about the environmental and economic benefits of device reuse can also foster greater acceptance and support for these practices within the patient community.
Future research should continue to evaluate the long-term effects of reusing various medical devices, diversifying the types of equipment studied, and extending the generalizability of findings to different clinical settings. Innovations in sterilization technology and new materials for medical devices might further enhance the feasibility and safety of reuse, advancing this cost- saving and eco-friendly practice.
Limitations
Limitations of the study include: the sample size, involving only 20 needles and four anesthetists, which may not broadly represent clinical diversity. Additionally, the study is confined to testing 20 reuse cycles, leaving the effects of further cycles unexplored. Examination of wear was primarily based on visible signs and electrical resistance, without delving deeply into potential microscopic damage or long-term effects of reuse in real clinical environments. Furthermore, the study did not evaluate direct patient outcomes or address how the reuse might be perceived by medical professionals and patients. To enhance reliability and applicability, future research could expand on these aspects, incorporating a larger and more varied sample, alternative sterilization techniques, and considerations of patient safety and acceptance.
Conclusion
The reuse of medical equipment, exemplified by our study on nerve block needles, offers significant economic and environmental benefits while maintaining high standards of patient safety. As healthcare systems worldwide strive to balance cost management with quality care, the development and implementation of effective reuse practices present a viable pathway to achieving these goals.
As more healthcare facilities consider adopting reuse strategies, the insights from empirical studies such as ours provide the groundwork necessary for informed decision- making. By fostering a culture of sustainability and safety, the medical community can leverage the reuse of equipment as a means of enhancing access to healthcare resources, reducing environmental impact, and ensuring patient safety remains paramount.
Overall, establishing comprehensive protocols and investing in education and training for healthcare providers are critical steps to enabling the successful integration of medical equipment reuse into modern medical practice. As we continue to explore the potential and challenges of this practice, collaboration between healthcare institutions, regulatory bodies, and academic researchers will be vital in optimizing reuse practices for future applications.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of the Institutional Review Board of Alanya Alaaddin Keykubat University (Date: 2022-05-13, No: 10354421)
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
References
-
Kantharia BK, Patel SS, Kulkarni G, et al. Reuse of explanted permanent pacemakers donated by funeral homes. Am J Cardiol. 2012. 2012;109(2):238-40.
-
Zamparas M, Kapsalis VC, Kyriakopoulos GL, et al. Medical waste management and environmental assessment in the Rio University Hospital, Western Greece. Sustain Chem Pharm. 2019;13:100163.
-
Aronson JK, Heneghan C, Ferner RE. Medical devices: definition, classification, and regulatory implications. Drug Saf. 2020;43:83-93.
-
Ross AS, Bruno MJ, Kozarek RA, et al. Novel single-use duodenoscope compared with 3 models of reusable duodenoscopes for ERCP: a randomized bench-model comparison. Gastrointest Endosc. 2020;91(2):396-403.
-
Aydogan E, Kozanhan B, Can S. Comparison of ultrasound images obtained by different disinfection methods used. Medicine (Baltimore). 2019;8(2):422-5.
-
Ai A, Anderson L, Safdar N, Shirley D. Barriers and facilitators to standardization of ultrasound use and probe disinfection in the ambulatory setting. Infect Control Hosp Epidemiol. 2020;41(4):469-71.
-
Baboudjian M, Gondran-Tellier B, Abdallah R, et al. Predictive risk factors of urinary tract infection following flexible ureteroscopy despite preoperative precautions to avoid infectious complications. World J Urol. 2020;38:1253-9.
-
La Fauci V, Costa GB, Facciolà A, Conti A, Riso R, Squeri R. Humidifiers for oxygen therapy: what risk for reusable and disposable devices? J Prev Med Hyg. 2017;58(2):E161.
-
Callahan A, Fries JA, Ré C, et al. Medical device surveillance with electronic health records. NPJ Digit Med. 2019;2(1):94.
-
Aydoğan E, Kozanhan B, Evaluation of ultrasonography probe disinfection habits in peripheral and/or central regional blocks applied with ultrasound guidance. Agri. 2019;31(1):9-14.
-
Tilleman K. Safety aspects of ultrasound scanning. Ultrasound in Assisted Reproduction and Early Pregnancy. London: CRC Press, 2020. p.252-9.
Figures

Figure 1. Measuring device (needles are marked with numbers and colored threads to distinguish them from each other)

Figure 2. Electrical resistance measuring equipment - measurement device has been shown, the red line in contact with needle tip and the black line, in contact on the stimulator connection point of the block needle
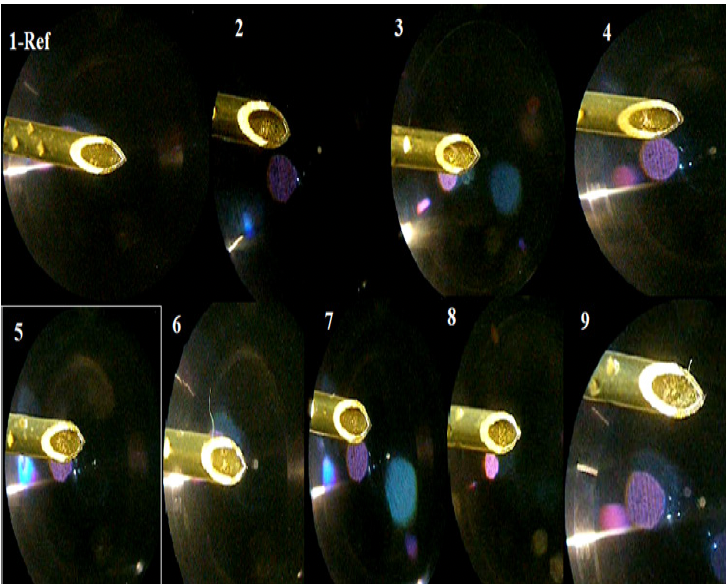
Figure 3. Numbers refer to needle number, number 1-ref; reference needle, number 2-3; (female) anesthetist who have experienced less than years, number 4-5; (male) anesthetist who have experienced less than years, number 6-7; (female) anesthetist who have five years or more experience, number 8-9; (male) anesthetist who have five years or more experience. (images have been obtained with 10x magnification of used needle tips)
Tables
Table 1. Mean load and electrical resistance of 20 block needles

Table 2. The load and electrical resistance values between the experiences upper 5 years and lower 5 years for female and male participants

Table 3. The load and electrical resistance values of whole data
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Eyup Aydogan, Betül Kozanhan, Yasin Tire, Munise Yıldız. In vitro assessment of wear in peripheral nerve block needles for re-use. Ann Clin Anal Med 2025;16(10):693-698

