Clinical and histological outcomes of entecavir and lamivudine in chronic hepatitis B therapy
Entecavir vs. Lamivudine: outcomes in chronic hepatitis B
- Received:
- January 16, 2025
- Accepted:
- March 19, 2025
- Published Online:
- March 28, 2025
- Printed:
- October 1, 2025
Authors
Abstract
Aim Chronic hepatitis B (CHB) is one of the major health concerns on the globe with an estimated 350 to 400 million cases and complications, including liver cirrhosis and hepatocellular carcinoma (HCC). The objective of this study is to assess the impact of entecavir and lamivudine treatment in the long-term viability of CHB patients, such as biochemical, serological, and histological outcomes.
Methods Data from a total of 75 patients diagnosed with chronic hepatitis B were taken. All the patients had received oral antiviral medications, including lamivudine or entecavir, for more than two years. The initial step included evaluation of clinical and lab parameters, which included serum levels of HBV DNA, ALT, HBeAg seroconversion, HBsAg histology, HAI score, and fibrosis score. The marks chosen for the study were 24,36, and 48 months. In-depth statistical analysis was then derived to study the effective treatment and compare the two moulds of treatment of CHB.
Results Entecavir demonstrated superior virological suppression, with 96.2%, 92.1%, and 100% of patients achieving HBV DNA levels <20 IU/mL at 24, 36, and 48 months, respectively, compared to 73.9%, 92.1%, and 90% in the lamivudine group. ALT normalization was observed in 90.5% of patients by the 48th month, with no significant difference between the two treatments. Both of the groups reported significant HAI lowering scores, i.e, a reduction of at least 2 throughout 36 moths with a composite improvement of at least 61.3 %. In comparison to this result, patients witnessing a reduction in fibrosis of a minimum of 1 point showed a minor decrease, which was not significant. All patients treated reported no signs of HBsAg clearance, while some were able to convert to anti-HBs.
Conclusion Entecavir has better virological effectiveness and similar biochemical and histological advantages in comparison to lamivudine on a long-term basis for converting patients with CHB. However, both regimens show only minimal reversal of fibrosis and are serologically unresponsive. This justifies the development of new treatment approaches such as combination therapies and immune modulators to facilitate performance improvements and fulfill unmet needs in the care of CHB.
Keywords
Introduction
Chronic Hepatitis B infection (HBV) is prevalent in 350-400 million people around the globe and continues to be a major contributing factor in liver diseases and death. 20-40% of individuals suffering from chronic HBV infection are additionally diagnosed with decompensated liver failure, cirrhosis, or hepatocellular carcinoma (HCC) 1,2. In consideration of these complications, the levels of viral replication are also significant. Patients that have an HBV DNA level less than (<300 copies/ml) are less likely to develop cirrhosis (4.5%) and HCC (1.3%). But in the cases for patients with high HBV DNA levels (≥106 copies/ ml), the possibility for them to develop cirrhosis increases to 36.2% while the possibility for HCC is at 15% 3,4.
The liver is one of the few organs in the body that is capable of regeneration to a remarkable extent; however, when this regenerative capacity is exceeded due to continuous injury to the liver, it results in the development of fibrosis that, over some time, leads to cirrhosis. It would be worthwhile pointing out that several recent studies have established that hepatofibrosis is reversible after the relevant cause for liver damage is removed. This treatment process is also dependent on antiviral therapy, which goes a long way in not only controlling the replication of the virus but also in ameliorating the histology of the liver, helping to slow down the development of fibrosis and associated long-term complications 1,5.
The therapeutic approaches currently employed in the management of HBV infections are classified into two principles, immune modulatory agents (for example, Peg-IFN-alpha) and orally active nucleos(t)ide analogs such as lamivudine, entecavir, and tenofovir. Nucleoside analogs are exceptionally successful in deterring the replication of viral DNA, normalizing aminotransferase (AT) levels, and averting complications 2,6. On the other hand, entecavir has greater resistance rates, which makes it a suitable option when managing patients long term. Data suggests that the patients start developing resistance to entecavir in under 1% after the 4-year mark and are nucleoside ignorant, in stark contrast with mono-resistant lamivudine patients who have a 40% resistance rate 1,3. Studies conducted after following certain steps conclude that as far as having renal failure involved, entecavir is much more suitable as compared to TDF 4,7.
The main purpose of this research is to compare entecavir and lamivudine long-term therapy in chronic hepatitis B (CHB) patients. The study pays careful attention to the impact of both therapeutic schemes upon virological, ALT normalization, histological (changes in HAI and fibrosis scores), and serological (HBsAg clearance and anti-HBs seroconversion) responses. It intends to assess the clinical efficacy of these alternatives to treatment and their input to the long-term management of patients. This research attempts to fill these clinical gaps by presenting evidence on the safety and efficacy of entecavir and lamivudine therapies.
This study focuses on the long-term effectiveness of entecavir and lamivudine about virological, biochemical, histological, and serological outcomes. The restricted gains achieved in current therapies are outlined by the limited serological responses like HBsAg clearance and anti-HBs seroconversion. The research was conducted in Turkey, which is an intermediate HBV endemic region, so the results help inform health policies as well as clinical practice at a more localized level using advanced strategies to treat CHB.
Materials and Methods
This retrospective study was conducted between 2000 and 2011 at the Gastroenterology Outpatient Clinic of Erciyes University Faculty of Medicine. The medical records of 150 patients diagnosed with chronic hepatitis B (CHB) were analyzed, excluding 47 patients due to insufficient follow-up data and 28 patients who had received antiviral treatments other than entecavir or lamivudine.
Inclusion criteria HBsAg positivity for at least six months, compensated liver disease (total bilirubin ≤2.5 mg/dL, INR ≤1.5, albumin ≥3.0 g/dL, and no history of variceal bleeding, hepatic encephalopathy, or ascites), baseline HBV DNA ≥104 copies/ mL, ALT levels between 1.3–10 times the upper limit of normal (ULN), and a minimum of two years of treatment with entecavir or lamivudine.
Exclusion criteria
Coinfection with HCV, HDV, or HIV, interferon therapy for more than 12 weeks, use of herbal treatments, or treatment with other oral antivirals.
Data collection
Demographic information, baseline and follow-up laboratory results (ALT, HBV DNA, HBsAg, HBeAg, anti-HBs, anti-HBe), and liver biopsy findings (fibrosis and histological activity index [HAI]) were analyzed over a minimum two-year period.
Evaluation criteria
HBV DNA suppression (<20 IU/mL), ALT normalization (<40 IU/mL), HBsAg loss, anti-HBs seroconversion, HBeAg seroconversion, and histological improvement (≥2-point reduction in HAI score, ≥1-point reduction in fibrosis score, or stabilization of fibrosis). Liver biopsies were assessed using the modified Knodell HAI and fibrosis scoring systems.
Statistical Analysis
All statistical analyses were conducted using SPSS version 20 for Windows XP. Results were expressed as mean ± standard deviation (SD) or median (25th–75th percentile). Comparisons of paired data were performed using the Wilcoxon signed-rank test, while categorical variables were analyzed using the Chi- square test and McNemar test. A p-value <0.05 was considered statistically significant.
Ethical Approval
This study was approved by the Ethics Committee of Erciyes University (Date: 2010-12-08, No: 2011/339).
Results
A total of 75 patients who received entecavir or lamivudine treatment for at least two years were included in the study. The mean age of the patients was 49±10.9 years, with 69.3% receiving entecavir and 30.7% receiving lamivudine. All patients were HBsAg-positive and anti-HBs-negative. At baseline, 14.7% of patients were HBeAg-positive, while 85.3% were anti-HBe- positive. Laboratory findings showed a median HBV DNA level of 2.62 × 106 IU/ml (1 × 104 - 5.62 × 107), an ALT level of 85 IU/ ml (61-149), a histological activity index (HAI) of 5 (4-6), and a median fibrosis score of 2 (1-3).
A significant decrease in HBV DNA levels was observed during the treatment period. At the 24th month, 89.3% of patients achieved HBV DNA levels below 20 IU/ml, with this rate remaining at 88.9% at the 36th month and increasing to 95.2% at the 48th month. Similarly, ALT levels showed marked improvement, with 68% of patients achieving ALT levels below 40 IU/ml at the 24th month, rising to 77.8% at the 36th month, and 90.5% at the 48th month (p < 0.05). Regarding serological parameters, 88% of patients were HBeAg-negative and anti- HBe-positive at the 24th month, increasing to 96.1% at the 36th month and 100% at the 48th month. However, no changes were observed in HBsAg or anti-HBs status, as all patients remained HBsAg-positive and anti-HBs-negative throughout the treatment period. A marked reduction in HBV DNA levels was noted, with 89.3% of patients achieving HBV DNA <20 IU/ ml at the 24th month, 88.9% at the 36th month, and 95.2% at the 48th month (p < 0.05). ALT levels also showed significant improvement, with 68% of patients achieving ALT levels <40 IU/ml at the 24th month, 77.8% at the 36th month, and 90.5% at the 48th month (p < 0.05).
The division of patients into two subgroups based on the use of entecavir or lamivudine revealed the results of liver biopsies performed at 24, 36, and 48 months of treatment, as detailed in Table 1.
When compared to pre-treatment levels, statistically significant biochemical improvements were observed across all treatment groups at 24, 36, and 48 months (p-values: 0.00, 0.00, and 0.00, respectively) (Table 1).
In terms of HBeAg seroconversion, only 18% (n = 2) of the 11 HBeAg-positive patients at baseline achieved HBeAg seroconversion at the 24th month, with no seroconversion observed at the 36th and 48th months (p > 0.05) (Table 2).
Histological activity index (HAI) improvements of ≥2 points were observed in 40% of patients at the 24th month, 61.3% at the 36th month, and 36.8% at the 48th month. These improvements were statistically significant, with p-values of 0.008, 0.000, and 0.045, respectively. Improvements in fibrosis scores of ≥1 point were observed in 24% of patients at the 24th month, 41.9% at the 36th month, and 31.6% at the 48th month. However, these changes were not statistically significant at any time point (p > 0.05) (Table 2).
Among patients who continued treatment for 24 months, 96.2% of those receiving entecavir and 73.9% of those receiving lamivudine achieved HBV DNA negativity (< 20 IU/ml). At the 36th month, this rate remained at 92.1% for both groups, while at the 48th month, 100% of entecavir-treated patients and 90% of lamivudine-treated patients achieved HBV DNA negativity (Table 3).
Similar improvements were observed in ALT levels. At the 24th month, ALT levels fell below 40 IU/ml in 73.1% of entecavir- treated patients and 56.5% of lamivudine-treated patients. At the 36th month, these rates were 78% and 75%, respectively. By the 48th month, ALT levels were within normal limits (< 40 IU/ml) in 90.9% of entecavir-treated patients and 90% of lamivudine-treated patients. These improvements in ALT levels were statistically significant in both groups (p < 0.05).
In histological evaluation, the rate of ≥2-point improvement in HAI scores was 38.9% in the entecavir group and 42.9% in the lamivudine group at the 24th month. At the 36th month, these rates increased to 64% and 50%, respectively. At the 48th month, 44.4% of entecavir-treated patients and 30% of lamivudine-treated patients achieved ≥2-point improvements in HAI scores. At the 36th month, this rate increased to 52% in the entecavir group and remained at 16.7% in the lamivudine group. At the 48th month, ≥1-point improvement in fibrosis scores was observed in 33.3% of entecavir-treated patients and 30% of lamivudine-treated patients. However, these changes were not statistically significant in either group (p > 0.05) (Table 3).
Discussion
The present study provides a comprehensive analysis of the long-term virological, biochemical, and histological outcomes of entecavir and lamivudine treatments in chronic hepatitis B (CHB) patients, contributing valuable insights into their comparative efficacy.
In both therapy groups, our data demonstrated noteworthy virological suppression, with 95.2% of patients achieving HBV DNA levels below 20 IU/ml at 48 months. Entecavir showed the best results in terms of virological efficacy, mainly at the earlier stages, with 96.2% patients from the entecavir group being HBV DNA negative at 24 months compared to 73.9% in the lamivudine group instead. This coincides with the results of Huang et al. 9 and Aljumah et al. 10, who noted the strong entecavir virological response in both the clinical trials and real life. Despite this, patients on lamivudine therapy had a higher frequency of virologic breakthroughs, consistent with Litwin et al. 2, who pointed out that the development of lamivudine resistance is a major drawback when used for a prolonged period.
Across all treatment groups, biochemical efficiency increased, particularly with normalising ALT levels. It was found that 90.6% of patients managed to keep their ALT levels below 40 IU/ml by the 48th month. These results are similar to those previously reported by Ozdemir et al. 11, who reported adequate control of ALT levels in subjects who were receiving long-term ALT therapy. Patients on entecavir did seem to have a slightly greater rate of ALT normalization than those on lamivudine in the early phases of treatment, although this study aligns with Gui et al. 1, who stated that entecavir-based regimens are more effective biochemically. By the 48th month of the study, the graphs presented are comparable, indicating the effectiveness of both techniques in the reduction of hepatic inflammation over time.
Data concerning histological outcomes showed improvement in the histological activity index (HAI) scores of the participants of the study, whereby there was a reduction of ≥2-point in 40%, 61.3%, and 36.8% of patients over 24, 36, and 48 months, respectively. These findings are indicative of histological improvements achieved through the use of antiviral therapies relevant to the research conducted by Gui et al. 1, which noted an improvement in histological features in patients receiving entecavir with combination therapy. On the other hand, the level of fibrosis reversal (≥1-point improvement and even lower was comparatively less and has not been found statistically significant) was found to be more moderate, which is in agreement with studies conducted by Çerçioğlu et al. 12 and Li et al. 6 regarding reversal of advanced fibrosis. Lamivudine was suggested by Hu et al. 13 to have greater histological reversibility if used in pediatric patients in comparison to adult patients. Primary etiology – advanced fibrotic noncirrhotic renovation (and decomposition) – warrants additional exploration.
Our study highlights the limited serological responses to antiviral therapy, particularly the lack of significant HBsAg and anti-HBs seroconversion. This aligns with findings by van Campenhout et al. 14 and Sarıkaya et al. 7, who emphasized that modern antiviral treatments do not fully eliminate CHB or prevent liver disease progression. In contrast, Li et al. 8 suggested that adjunctive therapies, such as immunomodulators or novel treatment approaches, may improve outcomes.
Concerns about antiviral toxicity remain relevant, especially in patients with coexisting HIV and CHB. While some studies, including Kara et al. 15 and Sarıkaya et al. 7, raised concerns about tenofovir-containing regimens, our findings suggest that entecavir remains a well-tolerated option. However, differences in monitoring and safety profiles across treatment regimens require further investigation.
Overall, our results support entecavir as a first-line therapy due to its superior virological and biochemical efficacy. However, the modest histological improvements and limited serological responses underscore the challenges in fully addressing CHB pathogenesis. These findings highlight the need for personalized treatment approaches, considering factors such as disease stage, patient age, and comorbidities, to optimize long-term outcomes.
Future steps in research should take a clinical perspective instead of focusing on particular medical strategies by evaluating combinational therapy methods and adjunctive medical strategies that bolster the overall outcome. In particular, these studies: Tatar et al. 16, Gui et al.1 imply that the combination of antiviral therapy with immune modulators or antifibrotic agents would result in a more profound method of treating CHB. But still, the lack of effective therapeutics for HBsAg clearance is a serious unresolved concern in combating CHB.
This study confirms the role of entecavir as a primary strategy while addressing CHB disease, especially during virological and biochemical optimization. Modification of therapeutic techniques would be facilitated by targeting wider therapeutic improvement since both histological change and surgical response proved to be minimal. There is a growing agreement in the literature and, in this sense, our results are consistent with that CHB can be more effectively combat for if it is in the patients’ best interests to customize the optimal approach to treatment; that the applications are continuously controlled and that combined therapies are also used.
Limitations
This study is limited by its retrospective design and the data collected within a specific period, which may affect the generalizability of the findings.
Conclusion
This study highlights the long-term efficacy of entecavir and lamivudine in virological suppression and biochemical improvement in chronic hepatitis B. Entecavir showed superior early virological response, reinforcing its role as a first-line therapy. Both treatments reduced ALT levels and hepatic inflammation, but fibrosis regression was limited, and HBsAg clearance remained unmet. These findings support individualized treatment strategies for long-term efficacy and safety. While entecavir remains a strong option, future research should explore combination therapies and immune modulators to improve histological and serological outcomes, aiming for better clinical results and functional cure rates.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of Erciyes University (Date: 2010-12-08, No: 2011/339)
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
References
-
Gui HL, Zhao CQ, Wang Y, et al. Histological outcome of Fuzheng Huayu plus entecavir combination therapy in chronic hepatitis B patients with significant liver fibrosis. J Clin Transl Hepatol. 2020;8(3):277.
-
Litwin M, Mikosińska A, Parys J, et al. Treatment of chronic hepatitis B: options, monitoring, special cases and adverse effects. J Educ Health Sport. 2025;77(1):1-19.
-
Chen H, Fu JJ, Li L, Wang X, Pan XC. Risk factors of low-level viremia in chronic hepatitis B patients receiving entecavir monotherapy: a retrospective cohort study. J Gastroenterol Hepatol. 2024;39(1):180-4.
-
Hsu YC, Chen CY, Tseng CH, et al. Antiviral therapy for chronic hepatitis B with mildly elevated aminotransferase: a rollover study from the TORCH-B trial. Clin Mol Hepatol. 2025;31(1):213-6.
-
Abayli B, Abaylı C, Gencdal G. Histopathological evaluation of long-term tenofovir disoproxil fumarate treatment in patients with hepatitis B e antigen- negative chronic hepatitis B. World J Gastrointest Pharmacol Ther. 2021;12(2):32.
-
Li M, Gao Y, Yang L, et al. Dynamic changes of cytokine profiles and virological markers during 48 weeks of entecavir treatment for HBeAg-positive chronic hepatitis B. Front Immunol. 2022;13:1-12.
-
Sarıkaya B, Çetinkaya RA, Yenilmez E, et al. Comparison of renal side effects of tenofovir disoproxil fumarate and entecavir treatments in patients with chronic hepatitis B infection: the results of five-year follow-up. South Clin Istanb Eurasia. 2022;33(4):138-44.
-
Li J, Dong XQ, Cao LH, et al. Factors associated with persistent positive HBV DNA levels in patients with chronic hepatitis B receiving entecavir treatment. Front Cell Infect Microbiol. 2023;13:1-11.
-
Huang YJ, Yang SS, Yeh HZ, Chang CS, Peng YC. Association of virological breakthrough and clinical outcomes in entecavir-treated HBeAg-positive chronic hepatitis B. PLoS One. 2019;14(8):e0221958.
-
Aljumah AA, Selayem NAB, Al-Howti SY, et al. Clinical and virological outcomes of entecavir therapy in patients with chronic hepatitis B: a real life experience. J Infect Chemother. 2019;25(1):12-6.
-
Ozdemir YE, Sahin Ozdemir M, Bayramlar OF, et al. Long-term follow-up of treatment-naïve HBeAg-negative patients with chronic hepatitis B. Ir J Med Sci. 2023;192(2):633-9.
-
Çerçioğlu D, Kınıklı S, Cesur S, Arslan K, Gönültaş M. Evaluation of histological response in chronic hepatitis B patients with tenofovir or entecavir therapy. Microbiology Bulletine. 2020;54(1):95-109.
-
Hu Y, Wu X, Ye Y, et al. Liver histological reversibility of lamivudine in treatment-naïve children with chronic hepatitis B: a retrospective cohort study from a single center Shanghai China. Transl Pediatr. 2023;12(6):1121.
-
Van Campenhout MJ, Brouwer WP, Xie Q, et al. Long-term follow-up of patients treated with entecavir and peginterferon add-on therapy for HBeAg- positive chronic hepatitis B infection: ARES long-term follow-up. J Viral Hepat. 2019;26(1):109-17.
-
Kara AV, Yıldırım Y, Ozcicek F, et al. Effects of entecavir, tenofovir and telbivudine treatment on renal functions in chronic hepatitis B patients. Acta Gastroenterol Belg. 2019;82(2):273-7.
-
Tatar B, Gül S, Köse Ş, Pala E. Long-term effects of tenofovir on liver histopathology in patients with chronic viral hepatitis B infection. Turk J Pathol. 2020;36(2):154.
Tables
Table 1. Liver biopsy findings of patients treated with entecavir or lamivudine at 24, 36, and 48 months

Table 2. Comparison of clinical and laboratory values of patients treated for 24, 36, and 48 months with baseline values
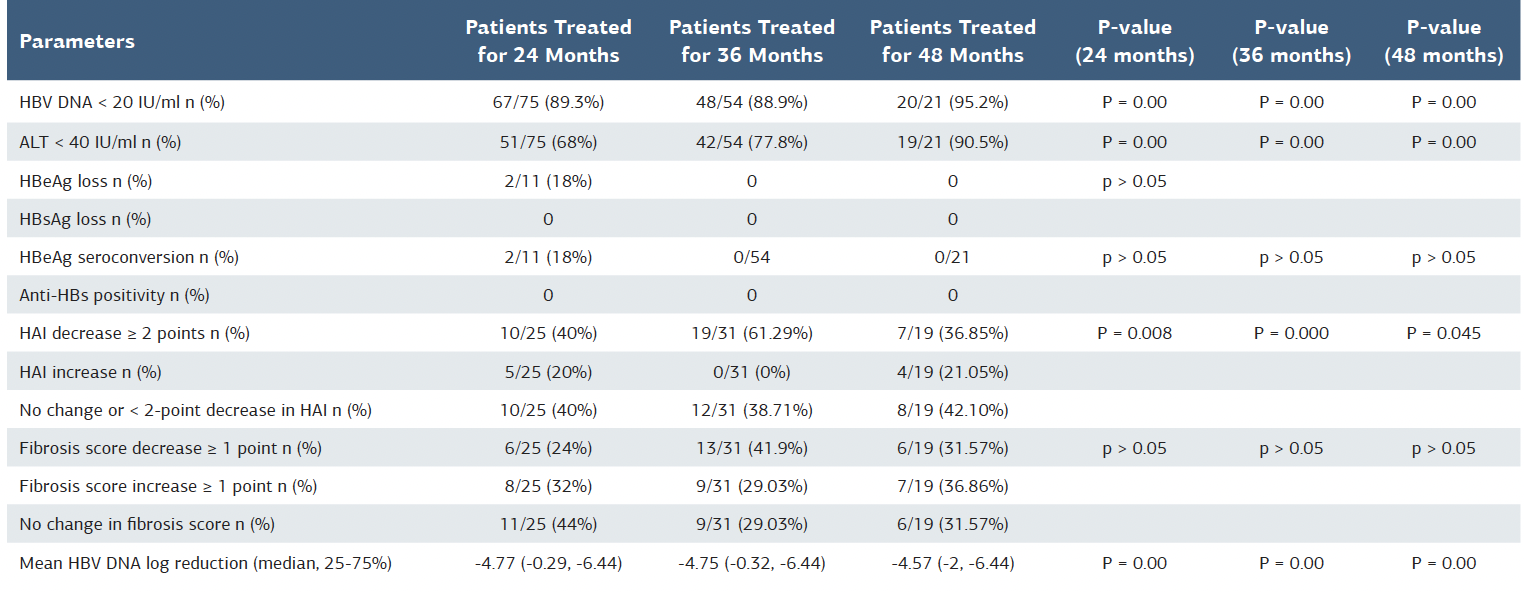
Table 3. Comparison of clinical and laboratory values of patients treated with entecavir and lamivudine at 24, 36, and 48 months with baseline values
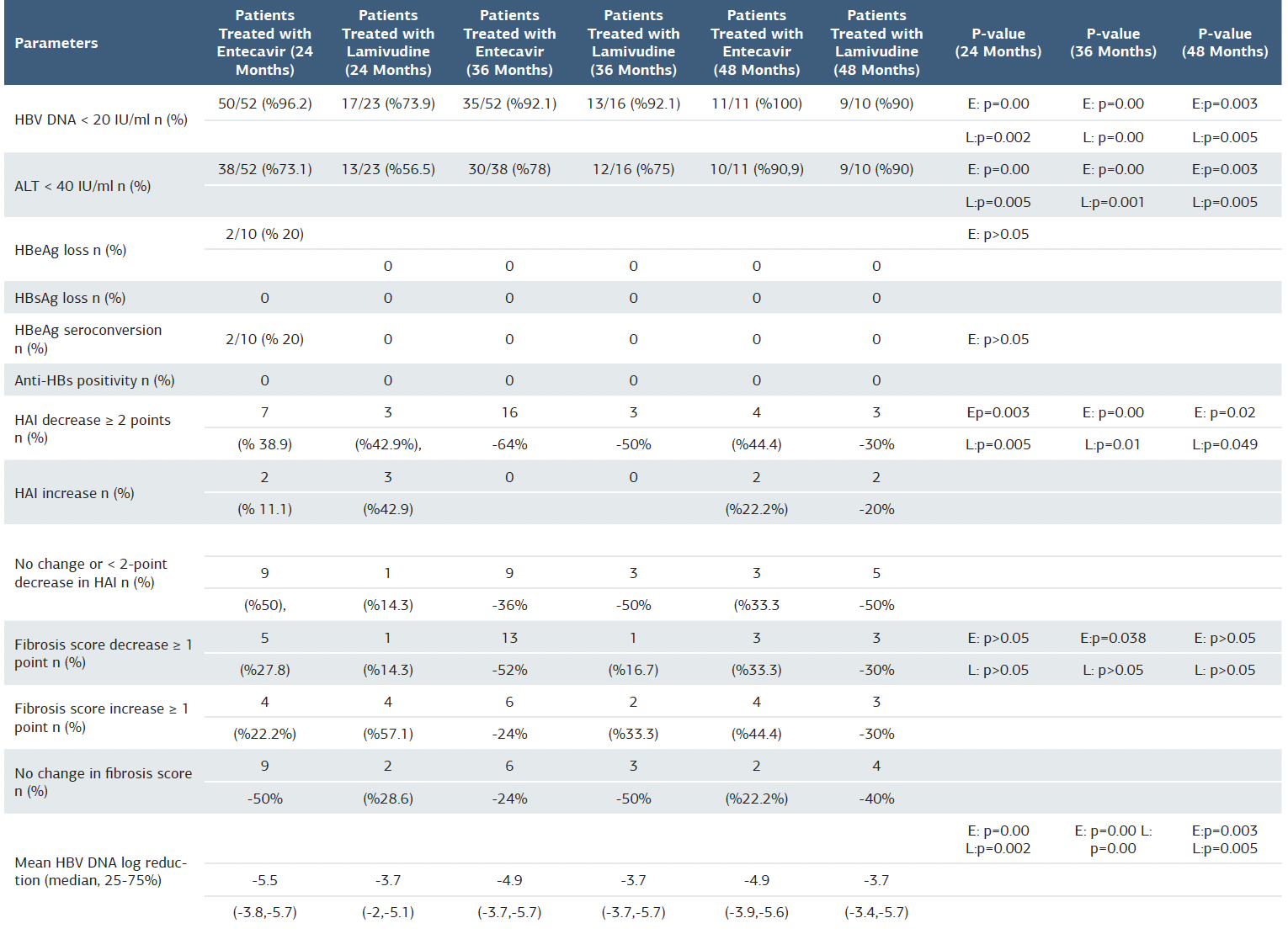
E = entekavir; L = lamivudin
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Neslihan Mandacı Şanlı, Mevlüt Başkol. Clinical and histological outcomes of entecavir and lamivudine in chronic hepatitis b therapy. Ann Clin Anal Med 2025;16(10):699-704

