The relationship between multi-inflammatory index and nondipping circadian pattern in patients with a new-diagnosis of essential hypertension
Multi-inflammatory index and non-dipper hypertension
- Received:
- February 4, 2025
- Accepted:
- March 3, 2025
- Published Online:
- March 17, 2025
- Printed:
- October 1, 2025
Authors
Abstract
AimPrevious studies showed that essential hypertension (EHT) had been related to chronic inflammation. A non-dipping circadian blood pressure (BP) pattern confers a greater cardiovascular morbidity and mortality. The multi-inflammatory index (MII) is a novel systemic inflammatory parameter. We aimed to assess the association between MII and the non-dipping BP pattern.
MethodsA total of 158 EHT patients were stratified into nondipper (n = 78) and dipper groups (n = 80) on the basis of 24h-ambulatory BP monitoring (24h-ABPM). 176 healthy subjects were recruited as control group. Data regarding 24h-ABPM, clinical, and laboratory findings were collected from the hospital records. MII was calculated as: Neutrophil count × C-reactive protein/Lymphocyte count.
ResultsMII was greater in the non-dippers as compared with the dippers and the controls [5.9(1.21-10.1), 3.32(1.34-8.87), and 1.79(0-4.1), respectively, p = 0.003]. MII was found to show correlation with age (0.200, p = 0.011), 24h-systolic BP 0.234, p = 0.003), daytime systolic BP (0.192, p = 0.016), nighttime systolic BP (0.277, p < 0.001), and LVMI (-0.195, p = 0.021). In the multivariate logistic regression model, daytime systolic BP [Odd ratio: 1.171(1.077-1.272, p < 0.001] and MII [Odd ratio: 1.150(1.029-1.284), p = 0.013] were found to be significantly associated with the non-dipping BP pattern. ROC curve analysis revealed a cut-off value of 2.01 with 71.1% sensitivity and 50.4% specificity for MII to predict the nondippers among the EHT patients.
ConclusionMII is significantly associated with a nondipping circadian BP pattern. MII could be a simple and useful marker to predict the nondippers among the EHT patients.
Keywords
Introduction
Hypertension continues to be a global issue, contributing significantly to morbidity and mortality. Although repeated blood pressure (BP) measurements in clinical settings have traditionally been the cornerstone for diagnosing essential hypertension (EHT), 24-hour ambulatory blood pressure monitoring (24-h ABPM) is now also used not only for diagnostic purposes but to reveal circadian patterns in hypertension. Normally, both systolic and diastolic blood pressures are expected to decrease by at least 10% during nighttime, a phenomenon known as the dipping blood pressure pattern. In contrast, when there is no such reduction of at least 10% of daytime BP readings during sleep, this is referred to as the non-dipping blood pressure pattern 1. Previous studies have demonstrated that a non-dipping BP pattern has been linked with a higher cardiac risk and increased all-cause mortality compared to the dipping BP pattern 2,3,4,5.
The multi-inflammatory index (MII) is a novel index encompassing C-reactive protein (CRP) and hematological parameters such as neutrophil and lymphocyte counts. Although initially proposed as a prognostic indicator in colorectal cancer, recent studies have also shown a significant relationship between MII and several cardiovascular diseases, such as atrial fibrillation, cerebrovascular events, and pulmonary embolism 6,7,8,9. However, its association with the circadian pattern of hypertension has yet to be elucidated.
The purpose of the present study is to determine the potential relationship between the novel MII and the dipping and non- dipping BP patterns in subjects with a new diagnosis of EHT.
Materials and Methods
This is a retrospective and cross-sectional study. The hospital’s digital database regarding the patients with a new diagnosis of EHT between October 2022 and May 2024 was interrogated. Records of 312 patients with a new diagnosis of hypertension were examined. 154 patients were excluded from the study based on our exclusion criteria, and finally, a total of 158 subjects with newly diagnosed EHT were enrolled. On the other hand, 176 age- and sex-matched normotensive healthy subjects were also recruited in our study to comprise the control group. Relevant demographic and clinical data of the subjects with a new diagnosis of EHT and the controls were obtained from digital hospital records. Exclusion criteria were set as: diabetes mellitus, known coronary artery disease, kidney failure, secondary hypertension, atrial fibrillation, active infection, malignancy, chronic rheumatic or inflammatory diseases, history of cerebrovascular events, and the use of steroids or anti-inflammatory medications. The patient group was then stratified into those with a dipping circadian BP pattern (the DG, n=78) and those with a non-dipping pattern (NDG, n=80) based on ambulatory blood pressure monitoring (ABPM) readings. The study was conducted by the standards set forth by the Helsinki Declaration.
At the office, a regular sphygmomanometer was used in each participant to measure the BP in the arm. Subjects were seated at least 10 minutes before the BP measurement, and 3 consecutive BP measurements were made at 5-minute intervals. Subjects with a mean office BP greater than 140/90 mm Hg (systolic or diastolic or both) were followed for twenty- four hours with ABPM (Mobil-O-Graph, Aachen, Germany). In daytime (07:00-23:00), the device performed BP measurements at 15-minute intervals, while in nighttime (23:00-07:00) at 30-minute intervals. After ruling out the secondary causes of hypertension, the diagnosis of EHT was made based on a 24- hour systolic BP mean >130 mm Hg/diastolic BP mean >80 mm Hg, a daytime mean systolic BP >135/diastolic BP >85 mm Hg, or a nighttime systolic BP mean >120 mm Hg/diastolic BP >70 mm Hg 13. A ≥10% reduction in daytime BP during the night was considered a dipping BP pattern. Patients without this 10% decrease were classified as non-dippers.
Echocardiographic assessment was conducted using the PowerVision 6000 (Toshiba, Tokyo, Japan). Simpson’s rule was utilized in the determination of the left ventricular ejection fraction (LVEF) 10. In each patient, LV mass index (LVMI) was calculated, and left ventricular hypertrophy was defined as LVMI >95 g/m2 for females and >115 g/m2 for males 10.
Blood samples were analyzed using an automatic CBC analysis device (Beckman Coulter Inc., CA, US). MII was calculated using the formula: Neutrophil count × C-reactive protein / Lymphocyte count 6.
Statistical Analysis
Statistical analysis was conducted using SPSS (Version 29.0 for Windows, IBM Corp., Armonk, NY, USA). Assessment of the distribution of quantitative data was made using the Kolmogorov-Smirnov test. Continuous data were expressed either as mean ± standard deviation or as median (25th and 75th interquartile range). On the other hand, categorical variables were expressed as counts (percentages). χ2 test, one- way ANOVA, and the Kruskal-Wallis H test were utilized based on data type. Multinominal logistic regression analysis was implemented to evaluate the associations of the variables with NDG. An analysis of the receiver operating characteristic (ROC) curve was implemented to delineate the cutoff of MII for NDG, if any. Any p-value <0.05 was accepted to be significant.
Ethical Approval
This study was approved by the Ethics Committee of Kırşehir Ahi Evran University Medical Faculty (Date: 2024-10-22, No: 2024-17/149).
Results
The groups were similar regarding gender, age, body-mass index, and baseline biochemistry parameters. The DG and NDG were similar in terms of smoking habits (p < 0.05); however, smoking was more common in the control group compared to the EHT patients (p < 0.001). The demographic and clinical variable of the subgroups were depicted in Table 1. MII was higher in the NDG compared with both the DG and controls [5.9 (1.21-10.1), 3.32 (1.34-8.87), and 1.79 (0-4.1), respectively, p = 0.003]. Neutrophil counts and hs-CRP levels were higher in the NDG than in both of the DG and control groups [for neutrophils 5610 (4200-6700), 4175 (2967-5577), and 4170 (3500-5300), respectively, p = 0.037; for hs-CRP 3 (1-6), 2.1 (1-3.4), and 1.75 (1-3), respectively, p = 0.006], while lymphocyte counts were higher in the DG and control group compared with the NDG [2155 ± 544, 2331 ± 918, and 2556 ± 844, respectively, p = 0.008].
LVMI was similar between the NDG and DG, but lower in the controls compared to the EHT patients [88.5 (78.4-105.6), 88.7 (79.7-106.4), and 84.2 (72.2-102.1), respectively, p = 0.013] (Table 1). Office systolic and diastolic BPs were similar between the dipper and non-dipper groups but lower in the controls compared to the EHT patients (p < 0.001). Ambulatory daytime and nighttime systolic and diastolic BPs were highest in the NDG and lowest in the controls (p < 0.001). While 24-hour systolic and diastolic BPs were similar between the DG and NDG, they were lower in the controls compared to the EHT patients (p < 0.001) (Table 1). Table 2 depicted the significant correlation of MII with LVMI (-0.195, p = 0.021), age (0.200, p = 0.011), 24-hour systolic BP (0.234, p = 0.003), daytime systolic BP (0.192, p = 0.016), and nighttime systolic BP (0.277, p < 0.001). Multinominal logistic regression identified daytime systolic BP [Odd ratio: 1.171 (1.077-1.272), p < 0.001] and MII [Odd ratio: 1.150 (1.029-1.284), p = 0.013] as significant predictors of the non-dipping BP pattern (Table 3). ROC curve analysis revealed a cutoff value of 2.01 with 71.1% sensitivity and 50.4% specificity for MII in predicting NDG among EHT patients (Figure 1).
Discussion
The key findings of our study may be summarized as follows: 1) compared with DG, MII is higher in NDG; 2) regression analysis revealed a significant association between MII and the non-dipping circadian BP pattern; and 3) ROC curve analysis demonstrated a cutoff value of 2.01 for MII in predicting non-dippers. The current study is the first to evaluate the relationship between MII and the non-dipping EHT, to the best of our knowledge.
Inflammation and the development of EHT are closely intertwined, leading to arterial wall inflammation and remodeling 11,12. For instance, depletion of B and T cells using mycophenolate mofetil has been shown to protect against hypertension and renal disease in animal models 11. Innate immune cells and endothelial cells express NLR- family pyrin domain-containing proteins NLRP1 and NLRP3, which activate inflammatory responses in the vessel wall through the release of interleukin (IL)-1β, high mobility group box 1 protein, and IL-18 13. Moreover, elevated levels of angiotensin II and endothelin 1 contribute to increased BP and vascular endothelial damage, leading to oxidative stress. This increased oxidative stress further triggers the T lymphocyte activation and the release of pro-inflammatory cytokines such as interferon-γ, Tumor necrosis factor-α, IL-17, and IL-6, hence creating a vicious cycle of inflammation 13,14,15. Kim et al. 16 found that alterations in circulating cytokine levels contributed to variations in BP. In a Japanese cohort, neutrophil counts were significantly associated with EHT risk in both sexes after adjusting for demographic factors 17. Similarly, we found that neutrophil counts were higher in NDG as compared with the DG and the control group.
The cardiovascular risk (CV) conferred by the non-dipping BP pattern has been greater compared to the dipping pattern due to more widespread end-organ damage, increased endothelial dysfunction, impaired diastolic left ventricular function, and chronic sympathetic nervous system activation 1. Coleman et al. 18 demonstrated significantly elevated central systolic BP and worsened central hemodynamics in non-dipper EHT patients, which may contribute to increased CV mortality. Akyüz et al. 19 reported higher neutrophil counts, neutrophil-to- lymphocyte ratios, and systemic inflammation-immune indices in non-dipper EHT patients compared to the DG. Sökmen et al. 1 found elevated levels of growth differentiation factor-15, an inflammatory cytokine, in patients with non-dipper EHT compared to those with a dipping BP pattern. Elçik et al. 20 observed faster coronary artery atherosclerotic deterioration in subjects with coronary artery disease who were in NDG.
In our study, MII was higher in non-dipper EHT patients, which is consistent with previous findings. Goztas et al. 21 demonstrated that MII could serve as a simple and useful mortality predictor in severe COVID-19 patients. Demirel et al. 8 reported that MII could indicate poor prognosis in ischemic stroke. Yüksel et al. 7 suggested that MII could predict new- onset atrial fibrillation in patients undergoing coronary bypass surgery. Doğanay et al. 22 found an association between higher MII, acute stent thrombosis, and early mortality in patients with acute coronary syndrome. Likewise, we found that MII was significantly associated with the non-dipping pressure rhythm, with a cutoff value of 2.01 as a simple and useful marker for predicting non-dippers among EHT patients.
Limitations
The relatively small sample size is a limitation of this study. Additionally, we did not assess the prognostic value of MII in non-dipper EHT patients. Lastly, the study was conducted at a single center, representing a relatively narrow patient population. Future studies with larger, multicenter cohorts are required to validate the results of our study.
Conclusion
This investigation demonstrated that MII was correlated with daytime, nighttime, and 24-hour systolic BP. Furthermore, MII was significantly associated with the non-dipping circadian pressure rhythm. MII could prove to be a simple and useful marker in the prediction of non-dipper EHT patients.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of Kırşehir Ahi Evran University (Date: 2024-10-22, No: 2024-17/149)
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
References
-
Sökmen E, Uçar C, Sivri S, et al. Association between growth differentiation factor 15 and non-dipping circadian pattern in patients with newly diagnosed essential hypertension. Med Princ Pract. 2019;28(6):566-72.
-
Brotman DJ, Davidson MB, Boumitri M, Vidt DG. Impaired diurnal blood pressure variation and all-cause mortality. Am J Hypertens. 2008;21(1):92-7.
-
Giles TD. Circadian rhythm of blood pressure and the relation to cardiovascular events. J Hypertens. 2006;24(Suppl.2):S11-6.
-
Mohammed AAS, Lin X, Yangyang Y, et al. The association of morning surge and night-time dipping blood pressure with significant and complex coronary artery lesions. High Blood Press Cardiovasc Prev. 2021;28(5):467-74.
-
Kalaycı B, Erten YT, Akgün T, Karabag T, Kokturk F. The relationship of age-adjusted Charlson comorbidity index and diurnal variation of blood pressure. Clin Exp Hypertens. 2019;41(2):113-7.
-
Agircan D, Bal M, Demir TG, Ethemoglu O. Multi-inflammatory index as a new predictive and prognostic marker of acute symptomatic seizures in patients with cerebral venous sinus thrombosis. J Stroke Cerebrovasc Dis. 2023;32(12):107453.
-
Yuksel A, Velioglu Y, Atasoy MS, et al. Multi-inflammatory index as a novel predictor of new-onset atrial fibrillation after off-pump coronary artery bypass grafting. Kardiol Pol. 2024;82(7-8):733-40.
-
Demirel ME, Akunal Türel C. The role of the multi-inflammatory index as a novel predictor of hospital mortality in acute ischemic stroke. Cureus. 2023;15(8):e43258.
-
Boyuk F. The role of the multi-inflammatory index as a novel inflammation- related index in the differential diagnosis of massive and non-massive pulmonary embolism. Int J Clin Pract. 2021;75(12):e14966.
-
Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28(1):1-39.e14.
-
Idris-Khodja N, Mian MO, Paradis P, Schiffrin EL. Dual opposing roles of adaptive immunity in hypertension. Eur Heart J. 2014;35(19):1238-44.
-
Agita A, Alsagaff MT. Inflammation, immunity, and hypertension. Acta Med Indones. 2017;49(2):158-65.
-
Aboukhater D, Morad B, Nasrallah N, et al. Inflammation and hypertension: Underlying mechanisms and emerging understandings. J Cell Physiol. 2023;238(6):1148-59.
-
Guzik TJ, Touyz RM. Oxidative stress, inflammation, and vascular aging in hypertension. Hypertension. 2017;70(4):660-7.
-
Sylvester MA, Brooks HL. Sex-specific mechanisms in inflammation and hypertension. Curr Hypertens Rep. 2019;21(7):53.
-
Kim KI, Lee JH, Chang HJ, et al. Association between blood pressure variability and inflammatory marker in hypertensive patients. Circ J. 2008;72(2):293-8.
-
Tomoyuki K, Maki M, Harumi U, Takako S. Neutrophil cl count is related to hypertension in workers: a cross-sectional study. Vascular Disease Prevention (Discontinued). 2007;4(3):225-8
-
Coleman CT, Stowasser M, Jenkins C, Marwick TH, Sharman JE. Central hemodynamics and cardiovascular risk in nondippers. J Clin Hypertens (Greenwich). 2011;13(8):557-62.
-
Akyüz A, Işık F. Systemic immune-inflammation index: a novel predictor for non-dipper hypertension. Cureus. 2022;14(8):e28176.
-
Elçik D, Duran M, Keleşoğlu Ş, et al. Effect of nondipper hypertension on coronary artery disease progression in patients with chronic coronary syndrome. Turk J Med Sci. 2021;51(3):1273-80.
-
Gozdas HT, Kayis SA, Damarsoy T, et al. Multi-inflammatory index as a novel mortality predictor in critically Ill COVID-19 patients. J Intensive Care Med. 2022;37(11):1480-5.
-
Doğanay B, Ozcan Celebi O. A novel inflammation indicator of acute stent thrombosis and in-hospital mortality in acute coronary syndrome: multiple inflammation index. Journal of Medicine and Palliative Care. 2023;4(2):168-75.
Tables
Table 1. Baseline demographic and clinical characteristics of the study population
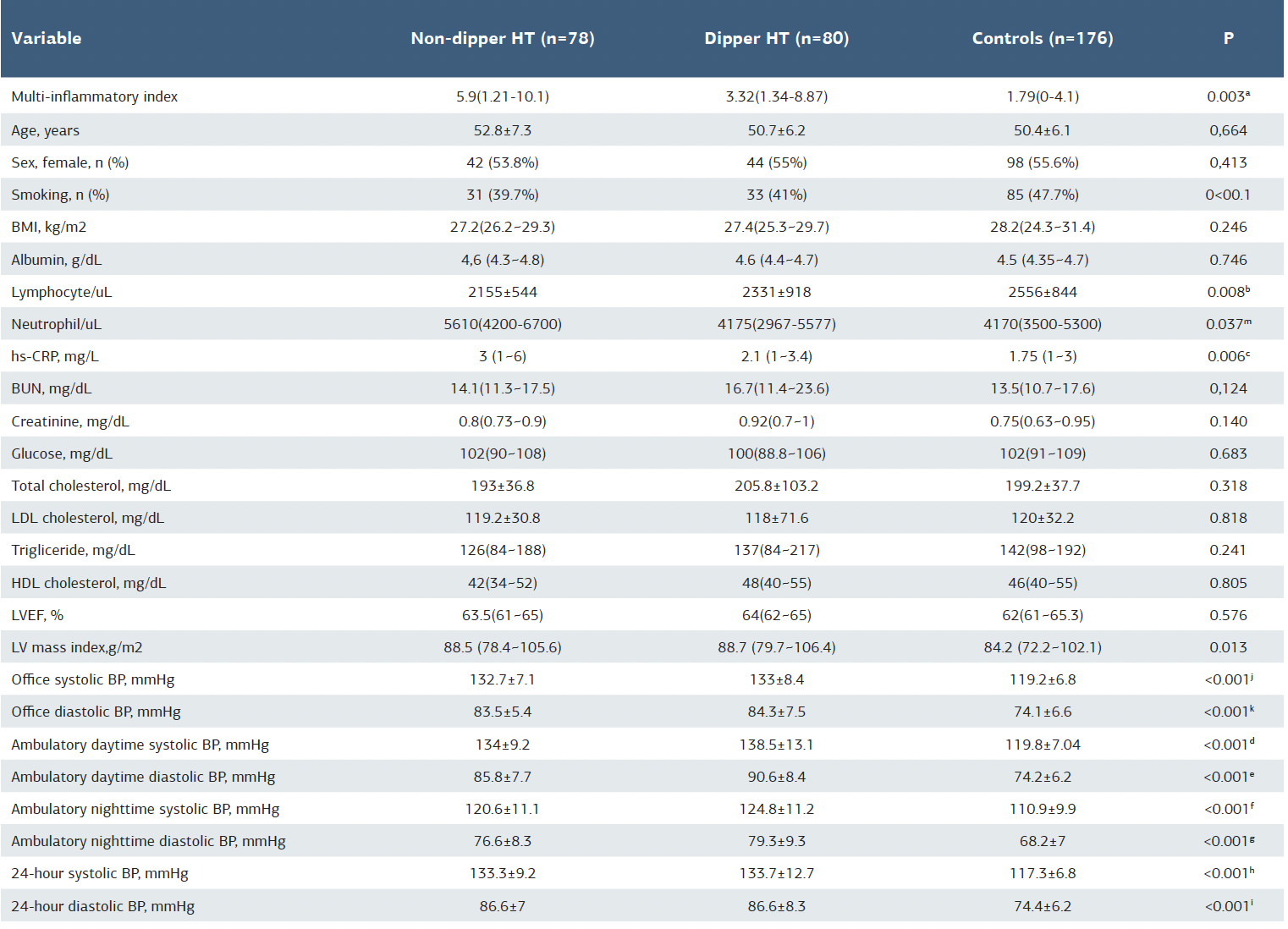
a p:0.009, between non-dipper and dipper hypertension groups; p<00.1, between non-dipper hypertension and control groups; p=0.021, between dipper hypertension and control groups. b p:0.585, between non-dipper and dipper hypertension groups; p:0.008 between dipper hypertension and control groups; p:0.202, between non-dipper hypertension and control groups. c p:0.121, between non-dipper and dipper hypertension groups; p:0.002, between dipper hypertension and control groups; p:0.112, between non-dipper hypertension and control groups. d p=0.010, between non-dipper and dipper hypertension; p<0.001 between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and control groups. e p=0.002, between non-dipper hipertension and dipper hypertension groups; p<0.001, between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and ocntrol groups. f p=0.788, between non-dipper hypertension and dipper hypertension groups; p<0.001, between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and ocntrol groups. g p=0.633, between non-dipper hypertension and dipper hypertension groups; p<0.001, between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and ocntrol groups. h p=0.579, between non-dipper hypertension and dipper hypertension groups; p<0.001, between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and ocntrol groups. i p=0.512, between non-dipper hypertension and dipper hypertension groups; p<0.001, between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and ocntrol groups. j p=0.538, between non-dipper hypertension and control groups; p<0.001, between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and dipper hypertension groups. k p=0.632, between non-dipper hypertension and control groups; p<0.001, between dipper hypertension and control groups; p<0.001, between non-dipper hypertension and dipper hypertension groups. m p=0.002, between non-dipper hypertension and control groups; p<782, between dipper hypertension and control groups; p<0.033, between non-dipper hypertension and dipper hypertension groups.
Table 2. Correlation of multiple immune-inflammation index with some study variables
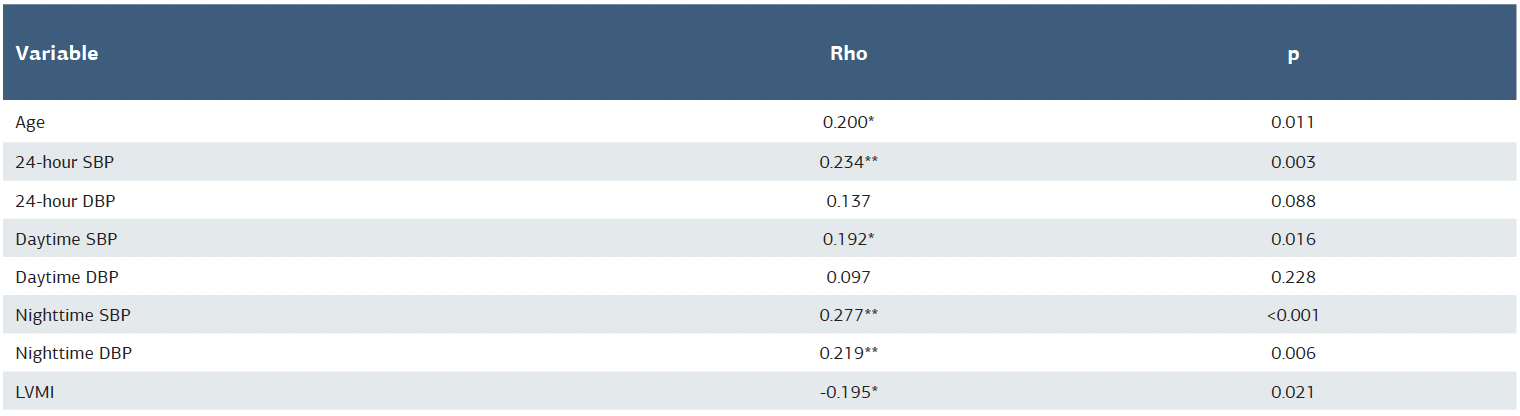
*p is significant at 0.05 level. **p is significant at 0.01 level.
Table 3. Logistic regression analysis showing the association of non-dipper hypertension with some study variables

Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Erdoğan Sökmen, Muhammet Salih Ateş. The relatieonship between multi- inflammatory index and nondipping circadian pattern in patients with a new- diagnosis of essential hypertension. Ann Clin Anal Med 2025;16(10):705-709

