Diagnostic and prognostic value of laboratory parameters in pediatric COVID 19 patients referred to intensive care unit by age groups
Lab markers in pediatric COVID-19 patients
- Received:
- March 25, 2025
- Accepted:
- May 12, 2025
- Published Online:
- August 10, 2025
- Printed:
- October 1, 2025
Authors
Abstract
AimThe aim of this study was to investigate the diagnostic and prognostic value of laboratory parameters according to age groups in paediatric COVID-19 patients referred to the intensive care unit.
MethodsThe study was conducted in Ankara Yenimahalle Training and Research Hospital Emergency Department COVID-19 Outpatient Clinic between 30 March 2020 - 1 June 2020. A total of 65 patients under the age of 18 years who were diagnosed with COVID-19 by RT-PCR test and later referred to the intensive care unit were evaluated. The patients were divided into four different groups according to age: 0-2 years (n = 22), 3-6 years (n = 9), 7-11 years (n = 13), and 12-18 years (n = 21). Demographic data, laboratory parameters, and clinical outcomes were analyzed retrospectively.
ResultsThere were significant differences in clinical outcomes between age groups (p<0.05). According to the ROC analysis results, CRP had the highest predictive value for clinical outcomes (AUC: 0.845, 95% CI: 0.730-0.961). When the cut-off value for CRP was set at 35.5 mg/L, clinical outcome could be predicted with 85.7% sensitivity and 78.2% specificity. WBC (cut-off value: 10.67×10^9/L) also had a significant predictive value (AUC: 0.811).
ConclusionIt was found that laboratory parameters in pediatric COVID-19 patients referred to intensive care differed according to age groups, and these parameters have an important role in the diagnosis and prognostic evaluation of the disease. In particular, CRP, WBC, lymphocyte, NLR, and MLR levels stand out as markers that can be used to predict the clinical course.
Keywords
Introduction
SARS-CoV-2 (severe acute respiratory syndrome coronavirus-2), which spread rapidly around the world and caused severe pneumonia, was declared a pandemic by the World Health Organization on 11 March 2000 1,3. Initially, it was seen more commonly in the adult population, but a significant increase in pediatric cases was observed in the later stages of the pandemic 3. Although COVID-19 infection in childhood is usually asymptomatic or mildly symptomatic, it can lead to more severe disease, particularly in children with underlying chronic diseases or in specific age groups 2,4.
The clinical and laboratory features of pediatric COVID-19 patients differ from those of adult patients 3,4. Reasons for the milder course of COVID-19 in children include developmental characteristics of the immune system, differences in angiotensin-converting enzyme 2 (ACE2) receptor expression levels, and fewer comorbidities 3,5. However, there are also significant differences in clinical and laboratory findings between different pediatric age groups.
Laboratory parameters play an important role in the diagnosis, severity assessment, and prognosis determination of COVID-19 infections. Although the laboratory findings associated with COVID-19 in adult patients are well-defined, there is limited data on the diagnostic and prognostic value of these parameters in the pediatric population especially in different age groups 4,5. Parameters such as white blood cell (WBC) count, lymphocyte count, neutrophil-to-lymphocyte ratio (NLR), platelet count, C-reactive protein (CRP), lactate dehydrogenase (LDH), ferritin, and D-dimer are associated with disease severity in patients with COVID-19 5,9.
Pediatric COVID-19 patients requiring intensive care may present with a more severe clinical picture and abnormal laboratory parameters. In order to develop appropriate treatment strategies and reduce mortality these patients, early diagnosis and identification of risk factors is very important 5,8. Especially in cases where PCR testing cannot be performed, or until the test results are available, evaluation of laboratory parameters can guide the clinician in the diagnosis and especially in the treatment approach 8,10.
The aim of this study was to investigate the diagnostic and prognostic value of laboratory parameters according to age groups in paediatric COVID-19 patients referred to the intensive care unit. It is our hope that the data obtained here will help clinicians in the management of paediatric COVID-19 patients and inform future studies.
Materials and Methods
Study Design and Participants
This study was conducted at Ankara Yenimahalle Training and Research Hospital Emergency Department Covid-19 Outpatient Clinic between 30 March 2020 and 01 June 2020. It consisted of patients under the age of 18 who came to Ankara Yenimahalle Training and Research Hospital Emergency Department Covid-19 Outpatient Clinic, were diagnosed with COVID-19 by reverse transcriptase polymerase chain reaction (RT-PCR) test and were later referred to intensive care unit (ICU) due to the need for a higher level of care. Case demographics, chronic diseases, emergency admission patterns, laboratory parameters, and patient outcomes were analyzed separately and collected retrospectively. In our hospital, patients presenting to the COVID-19 emergency department undergo a routine physical examination, and a haemogram and biochemical parameters are requested from each patient. Routine laboratory parameters, including haemogram results, were obtained from the first blood sample taken after the patients were admitted to the ED. PLR (platelet-to-lymphocyte ratio) was calculated as platelet count divided by lymphocyte count, MLR (monocyte- to-lymphocyte ratio) as monocyte count divided by lymphocyte count, and NLR (neutrophil-to-lymphocyte ratio) as neutrophil count divided by lymphocyte count. Patients under 18 years of age presenting to the COVID-19 emergency department undergo routine assessment and are referred to intensive care if they have a severe illness, including fever, dyspnoea, and/or chest imaging consistent with SARS-CoV-2 pneumonia, or new or increased need for supplemental oxygen and/or ventilation support; or critical illness, including respiratory failure requiring mechanical ventilation, acute respiratory distress syndrome, shock or systemic inflammatory response syndrome and/ or multi-organ failure. Patients were divided into four groups according to age: 0-2 years (infancy), 3-6 years (primary childhood), 7-11 years (secondary childhood) and 12-18 years (adolescence). Those under 18 years of age, diagnosed with COVID-19 by RT-PCR, referred to intensive care unit, and with complete data were included in the study. Cases over 18 years of age, diagnosed with COVID-19 by RT-PCR and not requiring intensive care, not diagnosed by RT-PCR, and missing any of the parameters studied were excluded from the study. The study was conducted by the tenets of the Declaration of Helsinki. Informed consent was not required due to the retrospective nature of the study.
Statistical Analysis
All statistical analyses were performed using SPSS version 20.0 for Windows. Descriptive statistics were used for the demographic analysis of patients. Chi-squared and Fisher exact tests were used to compare rates of categorical variables. Numerical values in the study data were expressed as mean ± standard deviation and minimum-maximum values. Kruskal- Wallis H test and Mann-Whitney U test were used in statistical evaluations according to the categorical (nominal or ordinal) and numerical independent group status of the statistically nonparametric variables. Based on the prognosis of adult and geriatric COVID-19 patients referred to the intensive care unit, laboratory parameters were evaluated with the receiver operating characteristics curve (ROC curve). When evaluating the ROC analysis, parameters with AUC<0.6 and not statistically significant (P > 0.05) were excluded. Results were evaluated at a significance level of p<0.05.
Ethical Approval
This study was approved by the Ethics Committee of Dr. Abdurrahman Yurtaslan Ankara Oncology Training And Research Hospital (Date: 2020-07-08, No: 2020/07.702).
Results
Out of 3470 patients under 18 years of age who presented to the COVID-19 emergency department, 65 patients who fulfilled the criteria were included in the study (Figure 1). The mean age of the patient groups was 0.8±0.8 for Group 1 (n = 22), 5± 0.8 for Group 2 (n = 9), 8.11± 2.3 for Group 3 (n = 13), 14.7± 2.1 for Group 4 (n = 21), and 54.5% (n = 36) of all patients were male. Patient demographics, length of ICU stay, clinical outcome, and use of mechanical ventilation are shown in Table 1. When evaluating the clinical outcome of the patients, there was a significant difference between groups (p<0.05). Rates for discharge from ICU were high in the 0-2 age group, decreased significantly in the 3-11 age group, and increased in the 12-18 age group. Although the rate of ventilation was similar between age groups, death was only observed in the 12-18 age group. Significant differences (p<0.05) were found between the groups in WBC, lymphocyte, platelet, platelet-lymphocyte ratio (PLR), NLR, MLR ratios, RDW, and CRP values (Table 2). According to the results of ROC analysis performed on the parameters that were significant in terms of clinical prognosis in pediatric COVID-19 patients, the parameter with the highest predictive value for clinical outcome was found to be CRP (AUC: 0.845, 95% CI: 0.730-0.961). When the cut-off for CRP was set at 35.5, it was predictive with 85.7% sensitivity and 78.2% specificity. WBC (cut-off: 10.67) also had a significant predictive value (AUC: 0.811). NLR and MLR had a lower predictive value (AUC: 0.713 and 0.689, respectively) (Table 3) (Figure 2).
Discussion
The diagnostic value of laboratory parameters in pediatric patients played an important role in early diagnosis and prognosis during the COVID-19 pandemic 2,5. In our study, there were significant differences in WBC, lymphocyte, platelet, PLR, NLR, MLR, RDW, and CRP levels between different age groups in pediatric COVID-19 patients admitted to the Intensive Care Unit (ICU). Our results suggest that CRP, WBC, NLR, and MLR are strong markers of clinical outcomes in pediatric COVID-19 patients.
Similar to our study, Perk et al. found that lymphopenia, anemia, and CRP elevation were observed in approximately 50% of pediatric COVID-19 patients 12. In addition, lymphopenia was observed in 65.4%, anemia in 48.1%, and thrombocytopenia in 27.2% of patients in their study. In a study by Demir et al, leukocyte and lymphocyte levels were found to be statistically significantly lower in healthy children than in pediatric patients diagnosed with COVID-19 (p = 0.01, p < 0.001) 5. In our study, upon examination of lymphocyte values according to age groups in pediatric COVID-19 patients, they were 6.4±4.2×109/L in the 0-2 age group, 3.1±1.1×109/L in the 3-6 age group, 3.1±1.1×109/L in the 7-11 age group, and 1.4±0.6×109/L in the 12-18 age group, and there was a significant difference between age groups (p < 0.05).
One of the important findings of our study was that WBC values showed a significant difference between age groups. WBC values were 16.6±12.7×109/L in the 0-2 age group, 8.6±3.6×109/L in the 3-6 age group, 5.8±2.4×109/L in the 7-11 age group and 9±6.1×109/L in the 12-18 age group (p=0.001). This is similar to the results of the study performed by Gumus et al. where they evaluated the clinical characteristics of pediatric COVID-19 infections 13. The aforementioned study found that leukocyte levels may be low, especially in the early stages of infection, and may come back to normal parameters in the later stages of the disease. Similar results were found in other studies that followed patients in pediatric intensive care units during the pandemic. Furthermore, investigating the role of laboratory parameters in the diagnosis of pediatric COVID-19 in the absence of PCR, Solmaz et al. found that WBC, neutrophil, platelet, and lymphocyte levels were significantly lower in patients than in the control group, and logistic regression analysis indicated that the best model was the one that included these parameters 11.
Platelet count and hematological parameters are important markers that can be used to predict the severity and prognosis of COVID-19 infections. In our study, the platelet count showed a significant difference between age groups (p<0.05) and was found to be 481±278×109/L in the 0-2 age group and 242±123×109/L in the 12-18 age group. Demir et al. found no significant change in platelet count in pediatric patients with COVID-19 5, whereas Solmaz et al. reported that platelet count was significantly lower in the patient group than in the control group 11. These different results suggest that COVID-19 infection shows a heterogeneous clinical manifestation in the pediatric age group and that laboratory parameters may vary depending on the age group.
In terms of inflammatory markers, CRP and procalcitonin levels were of particular note in our study. In the pediatric age group, CRP levels ranged from 13.6±55 to 17.8±14 mg/ dL and a significant difference was found between the groups (p = 0.005). A study performed by Xia et al. in pediatric COVID-19 patients reported that CRP levels were elevated in 45% of their patients 5. In addition, many studies have reported that CRP is elevated in pediatric COVID-19 patients 3,6,7,8,9,10,11,13. In our study, elevated CRP was an important laboratory finding in pediatric patients diagnosed with COVID-19. The predictive value of CRP was determined to be 35.5 mg/L by ROC analysis, and it was found to be a significant marker in terms of clinical course with a sensitivity of 85.7% and specificity of 78.2%. This suggests that CRP is an important marker of clinical severity and prognosis in pediatric COVID-19 patients.
Another notable finding in our study was that NLR (neutrophil/lymphocyte ratio) and MLR (monocyte/lymphocyte ratio) values differed significantly between age groups. NLR values were 1.6±2 in the 0-2 age group and 2.5±2.2 in the 12-18 age group (p=0.04). MLR values were 0.16±0.14 in the 0-2 age group and 0.40±0.29 in the 12-18 age group (p=0.001). Liu et al. reported that NLR was an important predictor of disease severity in COVID-19 patients and that high NLR values were associated with poor prognosis 15. Seyit et al. also found that NLR, MLR, and PLR could be used to predict the severity of COVID-19 infections 16. Similar to these studies, we also found that NLR and MLR had a high predictive value for clinical outcomes in paediatric COVID-19 patients (AUC: 0.713 and 0.689, respectively).
D-dimer elevation is considered an indicator of coagulopathy and thromboembolic complications in COVID-19 infections. In our study, D-dimer levels were 1304±267 ng/mL in the 0-2 age group, 255±311 ng/mL in the 3-6 age group, 92±137 ng/mL in the 7-11 age group and 509±907 ng/mL in the 12-18 age group. Although no statistically significant difference was found between the groups (p=0.260), it is noteworthy that D-dimer levels were particularly high in the 0-2 age group. Wang et al. reported that D-dimer elevation was observed in 51.5% of pediatric COVID-19 patients 17. Similarly, D-dimer elevation was found in a significant proportion of patients in the study by Perk et al 12. These findings suggest that the coagulation system may be affected in pediatric COVID-19 patients and that this may be related to the severity of the disease.
Limitations
Our study has several limitations. First, the study was conducted with single-centre data and the results may not be generalised to the whole population; second, some clinical data may not have been fully evaluated due to the retrospective design of our study. Finally, data from pediatric COVID-19 patients and healthy pediatric patients were not compared.
Conclusion
It was found that laboratory parameters in pediatric COVID-19 patients admitted to the ICU differed depending on the age group and that these parameters play an important role in the diagnosis and prognostic assessment of the disease, in particular CRP, WBC, lymphocyte, NLR, and MLR levels stand out as markers that can be used to predict the clinical outcome. Interpreting these parameters alongside clinical assessment is thought to help determine disease severity and prognosis. It will be useful to confirm these findings in future prospective multicentre studies with large patient populations.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of Dr. Abdurrahman Yurtaslan Ankara Oncology Training And Research Hospital (Date: 2020-07-08, No: 2020/07.702)
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
References
-
Chan JF, Yuan S, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395(10223):514-23.
-
Ludvigsson JF. Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults. Acta Paediatr. 2020;109(6):1088-95.
-
Dong Y, Mo X, Hu Y, et al. Epidemiology of COVID-19 among children in China. Pediatrics. 2020;145(6):e20200702.
-
Henry BM, Lippi G, Plebani M. Laboratory abnormalities in children with novel coronavirus disease 2019. Clin Chem Lab Med. 2020;58(7):1135-8.
-
Demir A, Gümüş H, Kazanasmaz H, Yükkaldıran A, Erat T. An examination of the laboratory data of paediatric COVID-19 cases. Int J Curr Med Biol Sci. 2021;1(2):33-7.
-
Lippi G, Plebani M. The critical role of laboratory medicine during coronavirus disease 2019 (COVID-19) and other viral outbreaks. Clin Chem Lab Med. 2020;58(7):1063-9.
-
Bulut B, Oz MA, Genc M, Yazici R, Mutlu H. The predictive value of hematologic parameters and inflammatory rates at admission on the need for hospitalization in COVID-19 patients over 65 years of age. Adv Health Sports Technol Sci. 2024;1(1):23-7.
-
Henry BM, Aggarwal G, Wong J, et al. Lactate dehydrogenase levels predict coronavirus disease 2019 (COVID-19) severity and mortality: a pooled analysis. Am J Emerg Med. 2020;38(9):1722-6.
-
Wang L. C-reactėve protein levels in the early stage of COVID-19. Med Mal Infect. 2020;50(4):332-4.
-
Cheng L, Li H, Li L, et al. Ferritin in the coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis. J Clin Lab Anal. 2020;34(10):e23618.
-
Solmaz A, Ercan TMF, Erat T, Solmaz F. The role of some parameters in diagnosis in the absence of PCR in children with COVID-19. Konuralp Med J. 2022;14(1):146-51.
-
Perk O, Ozcan S, Emeksiz S, et al. Clinical characteristics and treatment of COVID-19 patients admitted to the pediatric intensive care unit in our center. Pediatr Pract Res. 2022;10(1):26-31.
-
Gumus H, Ozcan Y, Kazanasmaz H, Demir A, Guzelcicek A. Clinical characteristics of COVID-19 infection in the pediatric age group. Electron J Gen Med. 2021;18(5):308.
-
Xia W, Shao J, Guo Y, Peng X, Li Z, Hu D. Clinical and CT features in pediatric patients with COVID-19 infection: different points from adults. Pediatr Pulmonol. 2020;55(5):1169-74.
-
Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019- nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63(3):364-74.
-
Seyit M, Avci E, Nar R, et al. Neutrophil to lymphocyte ratio, lymphocyte to monocyte ratio and platelet to lymphocyte ratio to predict the severity of COVID-19. Am J Emerg Med. 2021;40:110-4.
-
Wang D, Ju XL, Xie F, et al. Clinical analysis of 31 cases of 2019 novel coronavirus infection in children from six provinces (autonomous region) of northern China. Chinese J Pediatr. 2020;58(4):269-74.
Figures
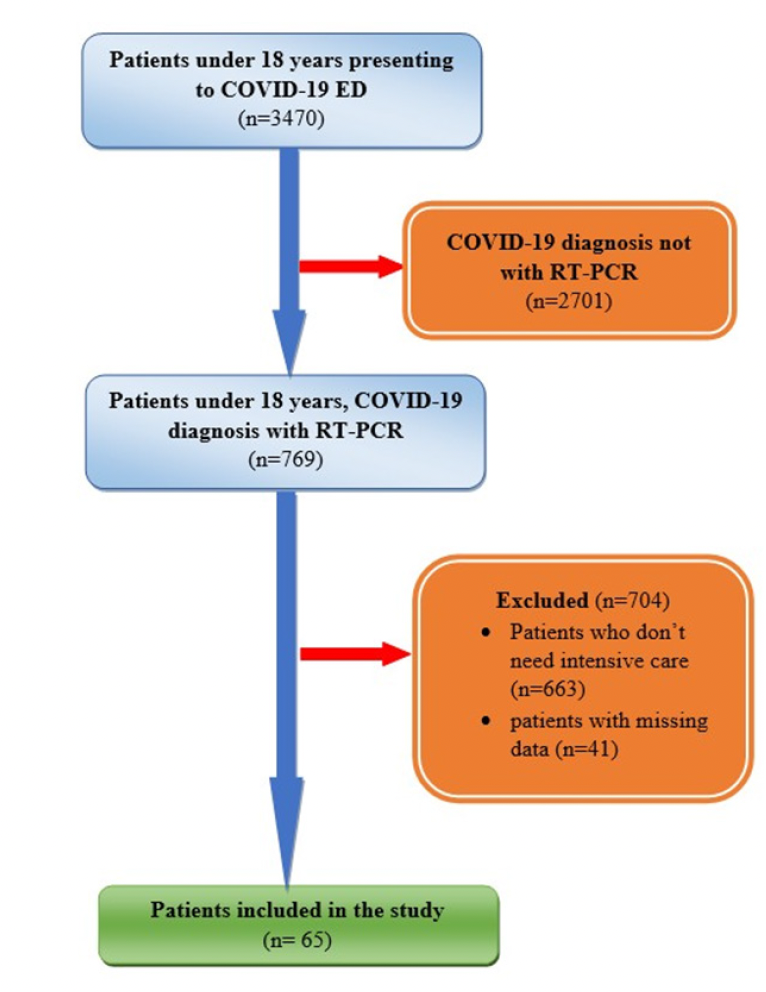
Figure 1. Workflow for pediatric COVID-19 patients
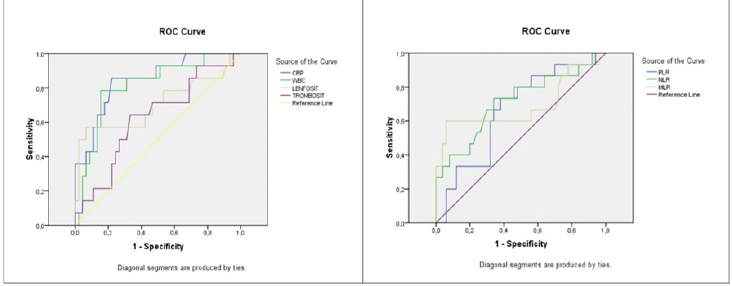
Figure 2. ROC curve of mortality assessment in COVID-19 patients aged 0-18 years
Tables
Table 1. Demographic data of the patients

As statistical analysis, Kruskal Wallis test and Chi-square test was used. * =p<0.05 was considered significant. SD = standard deviation
Table 2. Comparison of laboratory parameters according to patient groups
WBC = white blood cell; NLR = neutrophil to lymphocyte ratio; PLR = platelet to lymphocyte ratio; MLR = monocyte to lymphocyte ratio; CRP = c-reactive protein; MPV = mean platelet volume. For statistical analysis, the Kryskal-Wallis test was used. * =p<0.05 was considered significant SD = standard deviation
Table 3. Evaluation of laboratory panels found significant in terms of clinical course in pediatric COVID19 patients according to age groups by ROC analysis
WBC = white blood cell; CRP = c-reactive protein; NLR = neutrophil to lymphocyte ratio; MLR = monocyte to lymphocyte ratio; CI = confidence interval (lower bound- upper bound). As statistical analysis, ROC analysis was used. * =p<0.05 was considered significant
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Bensu Bulut, Murat Genç, Medine Akkan Öz, Mustafa Önder Gönen, Ramiz Yazıcı, Dilek Atik, Hüseyin Mutlu, Zekeriya Uykan. Diagnostic and prognostic value of laboratory parameters in pediatric COVID 19 patients referred to Intensive care unit by age groups. Ann Clin Anal Med 2025;16(10):715-719

