The relationship between depression, anxiety, and alexithymia in Huntington’s disease
Alexithymia in Huntington’s disease
- Received:
- July 2, 2025
- Accepted:
- August 4, 2025
- Published Online:
- August 18, 2025
- Printed:
- October 1, 2025
Authors
Abstract
AimNumerous studies have been conducted on psychiatric complaints such as depression, anxiety, and apathy in HD; however, due to the scarcity of studies examining alexithymia and their conflicting results, this study was deemed necessary. It was aimed to demonstrate alexithymia and the presence of difficulties in recognising and expressing emotions and extroverted thinking, which are subgroups of alexithymia, in patients with HD.
MethodsSixteen HD patients and sixteen healthy controls were assessed using the Beck Depression Inventory (BDI), Beck Anxiety Inventory (BAI), and the Toronto Alexithymia Scale-20 (TAS-20). Both groups were compared, including the TAS-20 subscales: TAS-A for difficulty identifying emotions, TAS-B for difficulty describing emotions, and TAS-C for externally-oriented thinking.
ResultsBDI, BAI, and TAS-20 scores were significantly higher in the patient group (P<0.02). Similarly, TAS-A, TAS-B, and TAS-C scores were also significantly higher in the patient group (P < 0.02). While there were 2 possible alexithymics in the healthy group, there were 6 possible alexithymics and 6 definite alexithymics in the patient group.
ConclusionIt has been shown that in HD, which has a high psychiatric burden, alexithymia and its subscales, difficulty in recognizing, expressing emotions, and
externally-oriented thinking, increase.
Keywords
Introduction
Huntington’s Disease (HD) is a progressive neurodegenerative disorder resulting from abnormal expansions of cytosine- adenine-guanine (CAG) repeats in the huntingtin gene (HTT) on chromosome 4p16, presenting with cognitive, motor, and neuropsychiatric symptoms 1. The neuropathological hallmark of HD includes the selective vulnerability of medium spiny neurons in the striatum, which make up over 90% of the cells, and the presence of intracellular aggregates known as inclusion bodies of mutant huntingtin protein (mHTT) 2,3. According to a meta-analysis of 33 studies published between 2010 and 2022, the incidence of HD was 0.48 per 100,000 cases 4. Although there is a juvenile form of the disease, it typically manifests in middle age, with an average onset age between 35 and 50 3. Motor symptoms seen in HD include choreiform movements, dysarthria, balance disorders, and parkinsonism. In HD, effects on the limbic system alongside the frontostriatal networks have been shown to cause cognitive changes 5. Emotions, sleep modulation, motivational behavior, memory, and social cognition, which are controlled by the amygdala, hippocampus, hypothalamus, and thalamus that constitute the limbic system, are also affected in HD 6. In HD, one can observe apathy, depression, generalized anxiety disorder, panic attacks, impulse control disorders, and psychosis 7. While motor symptoms are the most noticeable, psychiatric symptoms often cause more distress to patients and their relatives 8.
Alexithymia is a multidimensional personality structure that includes difficulty in recognising and defining emotions, difficulty in distinguishing emotions from somatic symptoms, extroverted thinking, and limitations in imagination 9,10. Individuals with high levels of alexithymia have difficulty perceiving emotions, identifying their emotions, and expressing their emotions to others. Along with the decrease in emotional attention, it leads to a decrease in memory and executive functioning. In individuals with high levels of alexithymia, a decrease in problem solving, tracking the information that needs to be carried out to solve the problem, synthesizing old and new information (updating), shifting between different mental tasks, and inhibiting dominant and automatic functions when necessary has been observed. All of these emphasize the importance of detecting the presence of alexithymia in the follow-up of HD patients 11. While alexithymia has been studied in many diseases, it has rarely been addressed in HD, which has such an intense psychiatric burden. Due to the different results in the few studies in the literature 12,13 and the lack of studies examining alexithymia in terms of difficulties in recognizing, expressing emotions, and externally-oriented thinking, this study was deemed necessary.
Materials and Methods
This study was conducted at the Neurology Outpatient Clinic Niğde Training and Research Hospital. Before the study, all participants were thoroughly informed about the study and provided written consent. The study was approved by the Non-Invasive Clinical Research Ethics Committee of Niğde Ömer Halisdemir University with protocol number 2024/90 on October 24, 2024. All procedures conformed to institutional or national ethical standards and the 1964 Declaration of Helsinki. Compliance with the ‘Regulation on Pharmaceutical Research’ (Official Gazette no. 27089, 23 December 2008).
Sixteen genetically diagnosed HD patients were included in the study. Sixteen healthy individuals, matched for age and sex, without psychiatric complaints, chronic neurological diseases, a family history of neurodegenerative diseases in first-degree relatives, or medication use, who came to the hospital as companions to patients, were selected as the control group. Patients with dementia, known psychiatric diagnoses, or those using antidepressants, antipsychotics, or anxiolytics were excluded from the study.
For both HD patients and the healthy control group, depression was assessed with the Beck Depression Inventory (BDI), anxiety with the Beck Anxiety Inventory (BAI), and alexithymia with the Toronto Alexithymia Scale-20 (TAS-20).
Beck Depression Inventory (BDI): This scale is used to assess the severity of depression. The reliability and validity of the Turkish version were carried out by Hisli Şahin in 1998 14. It consists of 21 items, each scored from 0 to 3; scores range from 0-9 for normal, 10-16 for mild depression, 17-29 for moderate depression, and 30-63 for severe depression.
Beck Anxiety Inventory (BAI): Used to measure anxiety severity, the validity and reliability study of the Turkish version was conducted by Ulusoy at al 15. It consists of 21 items, each scored from 0 to 3; 0-7 points indicate normal, 8-15 points indicate mild anxiety symptoms, 16-25 points indicate moderate anxiety symptoms, and 26-63 points indicate severe anxiety symptoms.
Toronto Alexithymia Scale-20 (TAS-20): This 20-item scale, developed by Bagby and colleagues, assesses alexithymia 16. The Turkish version’s validity and reliability were studied by Güleç et al. 17. Questions 4, 5, 10, 18, and 19 are reverse- scored. A TAS score ≤51 indicates no alexithymia, 52-60 suggests possible alexithymia, and ≥61 indicates definite alexithymia. TAS-A, composed of questions 1, 3, 6, 7, 9, 13, and 14, measures difficulty in identifying emotions; TAS-B, composed of questions 2, 4 (reverse), 11, 12, and 17, measures difficulty in describing emotions; TAS-C, composed of questions 5, 8, 10, 15, 16, 18, 19, and 20, provides insight into externally- oriented thinking.
Statistical Analysis
For statistical analysis, SPSS 26.0 (IBM SPSS Statistics 26 software; Armonk, NY: IBM Corp.) was used. The normality distribution of the data was evaluated using the Shapiro-Wilks test and histograms. Numerical variables were presented as mean ± standard deviation, and categorical variables as number (n) and percentage (%). The Chi-Square test was used to compare categorical variables between groups, with the Likelihood Ratio test preferred when expected cell frequencies were low. The Independent Samples t-test was used to compare age, depression, total alexithymia, TAS-B, and TAS-C scores between the HD patient and control groups, while the Mann -Whitney U test was used for anxiety and TAS-A scores. Spearman correlation analysis was applied to examine the relationship between the age and disease duration of HD patients with depression, anxiety, and alexithymia. Correlation coefficients were interpreted as follows: 0.00-0.25 = very weak, 0.26-0.49 = weak, 0.50-0.69 = moderate, 0.70-0.89 = high, and 0.90-1.00 = very high. The significance level was accepted as p<0.05.
Ethical Approval
This study was approved by the Ethics Committee of Nigde Ömer Halisdemir University (Date: 2024-10-24, No:2024/90).
Results
The study included 16 HD patients (7 women, 9 men) and 16 controls with similar demographic characteristics (9 women, 7 men). The mean age of HD patients was 58.62 ± 13.00 (minimum 26, maximum 77), while that of the control group was 51.50 ± 10.35 (minimum 26, maximum 70) (p=0.097). The disease duration was 10.50 ± 8.77 years. Depression, anxiety, and alexithymia levels of the patient and control groups were compared, as shown in Table 1, where BDI, BAI, and TAS-20 scores were significantly higher in the patient group. According to BDI, mild depression was observed in 6, moderate depression in 3, and severe depression in 6 of the HD patients, and mild depression was observed in 7, and moderate depression was observed in 2 of the healthy control group. Based on BAI, 6 of the patients with HD had mild, 6 had moderate, and 3 had severe anxiety, whereas 5 of the healthy control group had mild, 6 had moderate, and 3 had severe anxiety. In the healthy control group, only 2 individuals had possible alexithymia according to TAS-20, while in HD, there were 6 patients with possible alexithymia and 6 with definite alexithymia. TAS-A, TAS-B, and TAS-C scores were higher in HD patients (p<0.02). As shown in Table 2, there was a positive correlation between disease duration and TAS-20 and TAS-A. There was a positive correlation between BDI and BAI with TAS-B.
Discussion
Depression, anxiety, and alexithymia were shown to be higher in HD patients compared to healthy individuals. Additionally, in the patient group, difficulties in identifying emotions, expressing emotions, and externally-oriented thinking, as assessed by TAS- 20, were also higher. There was a positive correlation between depression, anxiety, and difficulty in expressing emotions.
In a study by Trinkler et al. involving 13 HD patients, no difference was found in TAS-20 scores between the patient and control groups, nor between emotions like anger, disgust, fear, happiness, surprise, and sadness. According to Trinckler et al., since emotion recognition and processing are similarly impaired in Parkinson’s disease and vascular lesions of the striatum, emotion recognition and processing may be similarly impaired in HD, which also progresses with striatal degeneration. In this study, the researchers thought that the scales may have been misleading even if alexithymia was present in patients with HD 18. In another study by Trinkler et al., involving 26 HD patients, alexithymia was not found to be increased 12. In our study, TAS-20 was significantly higher. Alt-hough neither study by Trinkler et al. showed an increase in alexithymia in HD compared to controls, a reduction in EEG activation during voluntary production of emotional facial expressions in patients with mild HD was demonstrated 12,18.
In Eddy et al.’s study using a 26-item TAS, scores of 62 or above were considered possible alexithymia, and 74 or above were considered definite alexithymia. Among 25 HD patients, 23 scored over 62, and 14 scored even higher. As in our study, the TAS score was higher in HD patients compared to the control group 13. Also, the TAS score was higher in manifest patients than in the pre-manifest group, which could support the increase in TAS-20 with disease duration observed in our study. A meta-analysis including sixteen studies indicated that the recognition of angry facial expressions was impaired in HD, and there was a tendency for the recognition of all negative emotions in both facial expressions and voice to be impaired 19. In Dupoux et al.’s study, HD patients had difficulty expressing emotions through both voice and language compared to healthy controls 20. Consistent with these studies, our study showed that HD patients have difficulty in recognizing and expressing emotions. The emotional recognition deficit in HD is partly explained by the general decline in cognitive and motor functions associated with the disease and partly by social-emotional deficits reflected in reduced eye tracking 21.
Although there are few studies on the prevalence of alexithymia in HD in the literature, it was stated in the review published by Cataine et al. that the frequency of depression and apathy correlated with emotional recognition deficit in Parkinson’s disease and HD. With disease progression, a decrease in emotional recognition and expression was observed. It has been observed that emotional recognition deficit, deterioration in interpersonal relationships, and social behavior problems 22. This was consistent with the positive correlation between disease duration and TAS-20 in our study.
In the study conducted by Tabrizi et al., the only cognitive impairment observed in presymptomatic individuals with HD genetic was found to be emotional recognition deficit. In the pre-HD period, the first complaint was observed as loss of empathy 23.
Limitations
The limitations of the study include the fact that it was conducted in a single centre, the small number of patients, the fact that the relationships between emotion recognition and facial expression recognition, and alexithymia were not examined, and that genetically diagnosed but presymptomatic individuals were not included in the study.
Conclusion
HD is a neurodegenerative disease with movement disorders, cognitive changes, and psychiatric symptoms. We aimed to highlight psychiatric symptoms, which might affect quality of life as much as motor symptoms, and to emphasize that these patients experience difficulties in recognizing, expressing, and externalizing emotions even before dementia develops.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of Nigde Ömer Halisdemir University (Date: 2024-10-24, No:2024/90).
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
References
-
Bates GP, Dorsey R, Gusella JF, et al. Huntington disease. Nat Rev Dis Primers. 2015;1:15005.
-
Ehrlich ME. Huntington’s disease and the striatal medium spiny neuron: cell-autonomous and non-cell-autonomous mechanisms of disease. Neurotherapeutics. 2012;9(2):270-84.
-
Tong H, Yang T, Xu S, et al. Huntington’s disease: complex pathogenesis and therapeutic strategies. Int J of Mol Sci. 2024;25(7):3845.
-
Medina A, Mahjoub Y, Shaver L, Pringsheim T. Prevalence and incidence of Huntington’s disease: an updated systematic review and meta-analysis. Mov Disord. 2022;37(12):2327-35.
-
Mason SL, Zhang J, Begeti F, et al. The role of the amygdala during emotional processing in Huntington’s disease: from pre-manifest to late stage disease. Neuropsychologia. 2015;70:80-9.
-
Mason SL, Schaepers M, Barker RA. Problems with social cognition and decision- making in Huntington’s disease: why is it important? Brain Sci. 2021;11(7):838.
-
Mühlbäck A, Hoffmann R, Pozzi NG, et al. Psychiatrische symptome der Huntington-krankheit [Psychiatric symptoms of Huntington’s disease]. Nervenarzt. 2024;95(9):871-84.
-
Beglinger L, O’Rourke JJ, Wang C, et al. Earliest functional declines in Huntington disease. Psychiatry Res. 2010;178(2):414-8.
-
Grigorescu C, Chalah MA, Ayache SS, Palm U. Alexithymie bei Multipler Sklerose-eine narrative übersicht [Alexithymia in multiple sclerosis - a narrative overview]. Fortschr Neurol Psychiatr. 2022;91(10):404-13.
-
Alimoradi Z, Majd NR, Broström A, et al. Is alexithymia associated with sleep problems? A systematic review and meta-analysis. Neurosci Biobehav Rev. 2022;133:104518.
-
Battista F, Lanciano T, Curci A. Does alexithymia affect memory for a crime? The relationship between alexithymia, executive functions, and memories. Front Psychol. 2021;12:669778.
-
Trinkler I, Devignevielle S, Achaibou A, et al. Embodied emotion impairment in Huntington’s Disease. Cortex. 2017;92:44-56.
-
Eddy CM, Rickards HE. Interaction without intent: the shape of the social world in Huntington’s disease. Soc Cogn Affect Neurosci. 2015;10(9):1228-35.
-
Hisli Şahin N. A study on the validity of Beck depression inventory in Turkish. Turkish J Psychol. 1988;7:3-13.
-
Ulusoy M. Şahin NH, Erkmen H. Turkish version of the Beck anxiety inventory: psychometric properties. J Cogn Psychother. 1998;12:163-72.
-
Bagby RM, Parker JD, Taylor GJ. The twenty-item Toronto Alexithymia Scale - I. Item selection and cross-validation of the factor structure. J Psychosom Res. 1994;38(1):23-32.
-
Güleç H, Kose S, Citak S, Yazici M. The Turkish version of the 20-Item Toronto Alexithymia Scale (TAS-20): reliability, validity, and factorial structure. Bull Clin Psychopharmacol. 2009;19(3):214-20.
-
Trinkler I, de Langavant LC, Bachoud-Lévi AC. Joint recognition-expression impairment of facial emotions in Huntington’s disease despite intact understanding of feelings. Cortex. 2013;49(2):549-58.
-
Henley SM, Novak MJ, Frost C, King J, Tabrizi SJ, Warren JD. Emotion recognition in Huntington’s disease: a systematic review. Neurosci Biobehav Rev. 2012;36(1):237-53.
-
Gallezot C, Riad R, Titeux H, et al. Emotion expression through spoken language in Huntington disease. Cortex. 2022;155:150-61.
-
Kordsachia CC, Labuschagne I, Stout JC. Visual scanning of the eye region of human faces predicts emotion recognition performance in Huntington’s disease. Neuropsychology. 2018;32(3):356.
-
Kordsachia CC, Labuschagne I, Stout JC. Beyond emotion recognition deficits: a theory guided analysis of emotion processing in Huntington’s disease. Neurosci Biobehav Rev. 2017;73:276-92.
-
Tabrizi SJ, Scahill RI, Owen G, et al. Predictors of phenotypic progression and disease onset in premanifest and early-stage Huntington’s disease in the TRACK-HD study: analysis of 36-month observational data. Lancet Neurol. 2013;12(7):637-49.
Tables
Table 1. Comparison of depression, anxiety, and alexithymia levels of participants
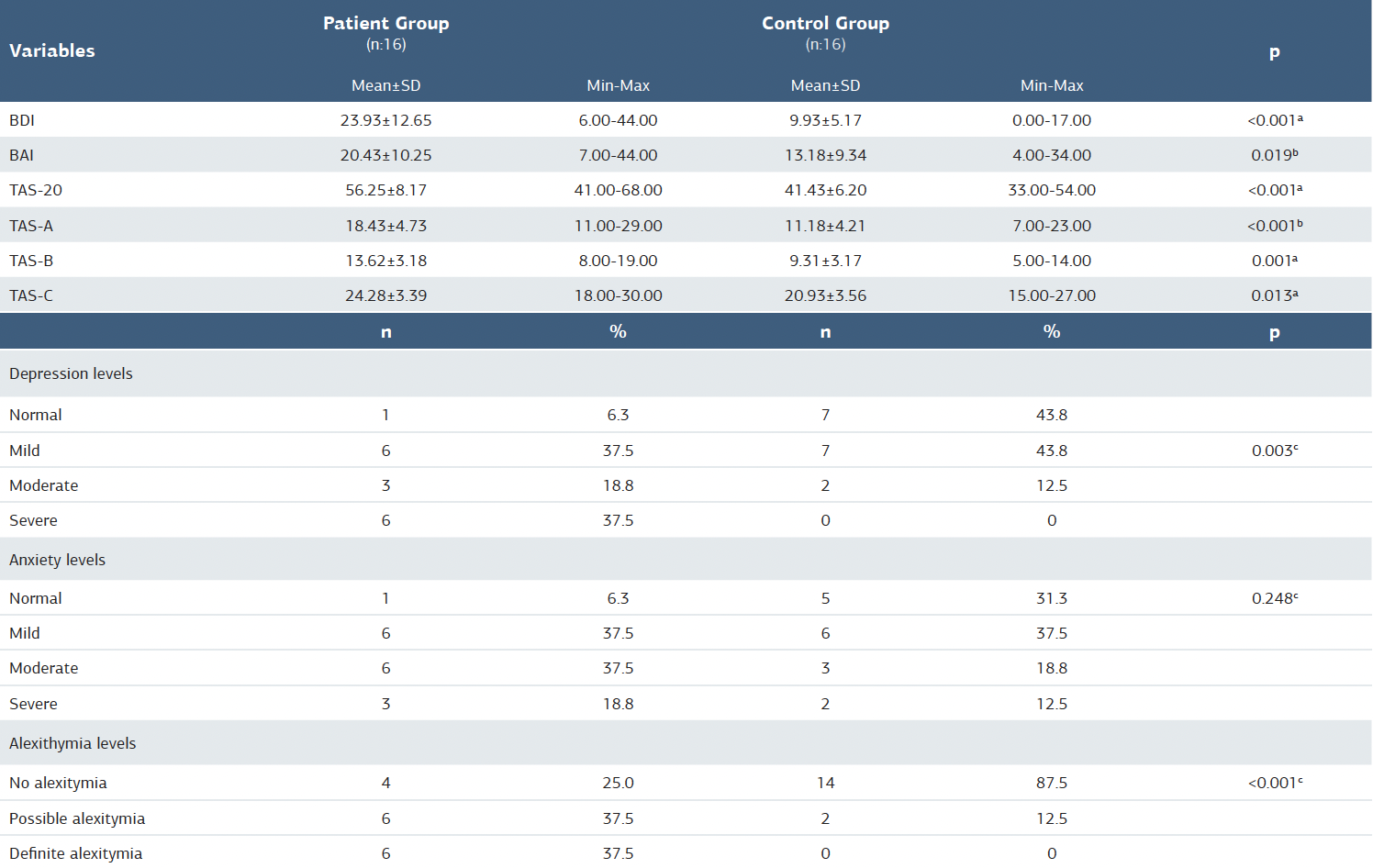
N = number; SS = standard deviation; min = minimum; max = maximum; % = percentage; BDI = Beck Depression Inventory; BAI = Beck Anxiety Inventory; TAS-20 = Toronto Alexithymia Scale-20; a = independent sample t-test; b = Mann-Whitney U test; c = likelihood ratio
Table 2. Examination of the relationship between Huntington’s disease, depression, anxiety, and alexithymia
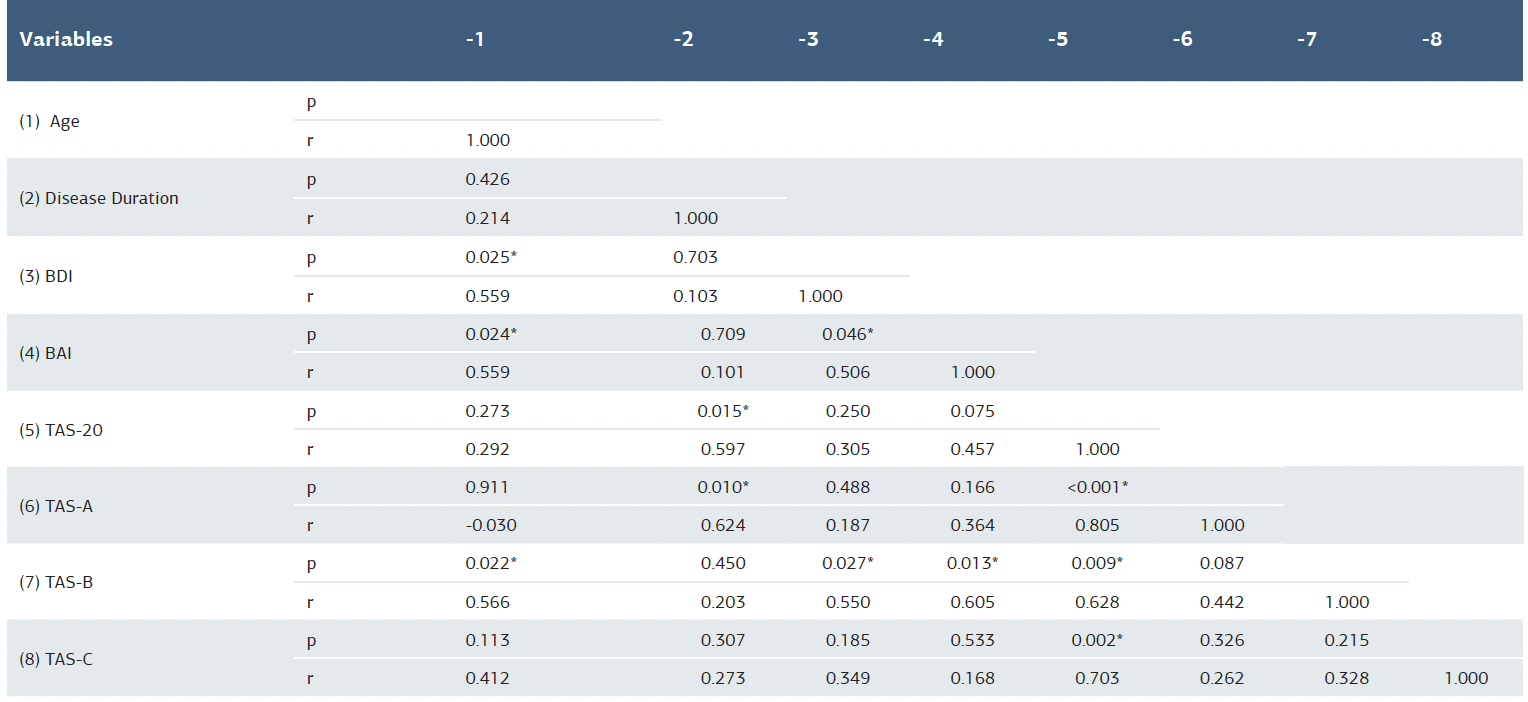
BDI = Beck Depression Inventory; BAI = Beck Anxiety Inventory; TAS-20 = Toronto Alexithymia Scale-20; * = Spearman's correlation coefficient
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Dilek İşcan, Huriye Ünlüel, Hilal Özgüner, Şengül Şenel. The relationship between depression, anxiety, and alexithymia in Huntington’s disease. Ann Clin Anal Med 2025;16(10):720-724

