Gut microbiota, diet, and metabolic diseases: a triangular relationship
Microbiota–diet–metabolism triangle
Authors
Abstract
Current evidence highlights the importance of the gut microbiota in connecting the metabolic responses of dietary interventions. A scoping review system- atically mapped and synthesized the evidence from 49 randomized controlled trials (RCTs) on the mediating effect of changes in gut microbiota. Databases were searched for RCTs involving adult participants that reported on both gut microbiota and metabolic outcomes. Eligible studies included defined dietary interventions. Interventions were found to alter microbiome diversity reliably and boost the prevalence of beneficial organisms (such as Bifidobacterium, Fae- calibacterium prausnitzii, and Akkermansia muciniphila). Improvements in insulin sensitivity, lipid profiles, inflammatory biomarkers, and hepatic fat content are often accompanied by such microbial alterations. Fifteen studies provided direct evidence that dietary modulation of the microbiota leads to quantifiable metabolic effects. Current RCT evidence supports the potential of dietary interventions to modulate the gut microbiota and promote metabolic health. Findings highlight the potential of microbiome-targeted nutritional interventions within precision medicine and public health policy, as well as the need for longer-term, mechanistically detailed, and geographically diverse trials.
Keywords
Introduction
BackgroundThe gut microbiota, a complex community of microorganisms residing within the human gastrointestinal system, is crucial for overall health and homeostasis. The gut microbiota plays a crucial role in multiple physiological functions, including digestion, the immune system, and neurological signaling. It has been emphatically established that gut microbiota plays a crucial role in controlling host metabolism, protecting against pathogens, and regulating the immune system.1,2
Diet is one of the predominant factors that influence the composition of gut microbiota. Nutrient intake is a source of nutrients upon which the microbiota are sustained, and it is well known that brief alterations in diet can cause demonstrable changes in microbiota composition. Indeed, dietary interventions have demonstrated that changes in macronutrient content lead to substantial modifications in the gut ecosystem within a short period, with dramatic changes in bacterial populations occurring within 24 hours.3 These alterations entail not only shifts in microbial composition but also in function, including the production of metabolites such as short-chain fatty acids (SCFAs), which are reported to be critical modulators of inflammation, lipid metabolism, and insulin sensitivity.
Recent evidence also emphasizes a strong association between host gut microbiota and metabolic disorders such as obesity, type 2 diabetes (T2D), and metabolic syndrome (MetS). The microbial balance, known as dysbiosis, is often disrupted in patients with metabolic diseases. These changes are characterized by derangement of intestinal barrier function, low-grade systemic inflammation, and disturbed energy metabolism.4 Furthermore, microbial metabolites, such as imidazole propionate, have been linked to the development of insulin resistance, suggesting a direct mechanistic relationship between microbial activity and the host’s metabolic health.
Together, these evidences highlight the role of gut microbiota as a mediator and a possible therapeutic target in the pathogenesis of metabolic disease. They also link diet to translationally important and modifiable features of microbial health, thereby connecting diets and their nutritional strategies with disease prevention and treatment.
Existing Research GapsAlthough there is an increasing body of literature on gut microbiota, dietary factors, and metabolic diseases, little attention has been paid to the interaction between these factors; most studies have considered them as independent or univariate parameters in the long term. For example, microbiota is described as being shaped by diet and as contributing to metabolic diseases through dysbiosis but is rarely discussed as one vertex of an entangled and dynamic triangle with nutrition and host metabolism. This fragmented strategy has so far hindered knowledge about the role of dietary components in shaping microbial communities and how this contributes to the development or aggravation of metabolic diseases. Furthermore, due to the absence of an integrative framework and a lack of comparison across dietary patterns, microbiota, and metabolism, a critical gap exists in the development of personalized nutritional strategies for optimal disease prevention. Accordingly, a comprehensive overview of the relationship between diet, gut microbiota, and metabolic health is warranted.
Rationale for Scoping ReviewThe complexity of interactions among gut microbiota, diet, and metabolic diseases demands a comprehensive and integrated view of the current literature. A weakness of conventional systematic reviews is the limitation to specific interventions or outcomes, which may miss overarching trends and contextual considerations. A scoping review is well suited to explore new, complex, or diverse bodies of evidence in which the evidence is heterogeneous concerning study design, population, and/or conceptual focus.5 A scoping review method promises to highlight research gaps, controversial points, the limitations of the concept in question, and the diversity of methodologies used to explore the complexity of the interaction among gut microbiota, diet, and metabolic diseases.
As the gut microbiota literature is also growing rapidly, a growing body of evidence is pointing to a unique yet interrelated role of diet within microbial ecosystems and vice versa, with microbial ecosystems affecting metabolic health-related conditions (i.e., obesity, diabetes, and cardiovascular disease).1 Yet, the three dimensions are rarely assessed simultaneously in an integrated fashion. Thus, in a scoping review employing the enhanced methodology proposed by Peters et al.,6 there is a critical need to systematically catalog triangular interactions, compare key findings, and identify gaps for cross-disciplinary integration. Such a framework is essential for informing future research directions, guiding clinical practice, and facilitating the development of targeted intervention strategies for dietary management and the prevention of metabolic disease.
Objectives and Research Questions1. To survey the current literature on the triangular connection of gut microbiota-diet-metabolic diseases.
2. To investigate how diet influences the gut microbiota.
3. To determine the mechanisms by which alterations in gut microbiota composition and function promote the development and progression of metabolic diseases.
4. To identify evidence gaps and recommend avenues for research.
To evaluate whether integrated approaches involving diet and microbiota can be effectively leveraged for metabolic disease prevention and management.
Materials and Methods
Protocol and RegistrationThis scoping review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) guidelines. No formal protocol was registered for this review. However, the methodology was developed in accordance with the frameworks established by Arksey and O’Malley.5 and further refined by Peters et al.6
Eligibility CriteriaThe inclusion criteria were:
• Peer-reviewed journal articles published up to and including May 2025.
• Studies written in English.
• Research involving only adult human subjects.
• Randomized controlled trials (RCTs) examining all three dimensions: diet, gut microbiota, and metabolic disease.
Excluded were:
• Non-English publications, pediatric-related studies, commentaries, editorials, and abstracts without a full text.
• Animal studies, in vitro studies.
The rationale was to include the most relevant, up-to-date, and scientifically rigorous evidence to comprehensively map the triangular relationship between diet, gut microbiota, and metabolic disease.
Information SourcesDatabases searched included:
• PubMed
• Web of Science
• Cochrane
The most recent search was conducted on May 3, 2025.
SearchThe search strategy involved keyword combinations such as (“gut microbiota” OR “microbiome”) AND (“diet” OR “nutrition”) AND (“metabolic syndrome” OR “obesity” OR “diabetes”). Boolean operators (AND/OR) and truncation (e.g., microb*) were used to optimize sensitivity and specificity. Search filters limited results to English-language articles published up to and including May 3, 2025.
Selection of Sources of EvidenceThe search yielded 2,615 articles from PubMed, Scopus, and the Cochrane Library. Rayyan Duplicate Detection identified 1065 duplicate records. Following manual screening, 541 duplicate articles were removed, 503 were resolved, and 21 were identified as non-duplicates, resulting in 1,550 records for screening.
Two reviewers independently conducted title and abstract screening using the Rayyan software. Conflicts were flagged by the system and resolved through discussion. Following the inclusion and exclusion criteria, 671 full-text articles were reviewed. Finally, 49 RCTs were identified that met the inclusion criteria and were included in the review.
Data Charting ProcessInformation was graphed using a pre-created extraction form, which was piloted and used by the review team to modify the list. Data were extracted independently by two reviewers in duplicate. Differing opinions were resolved by consensus.
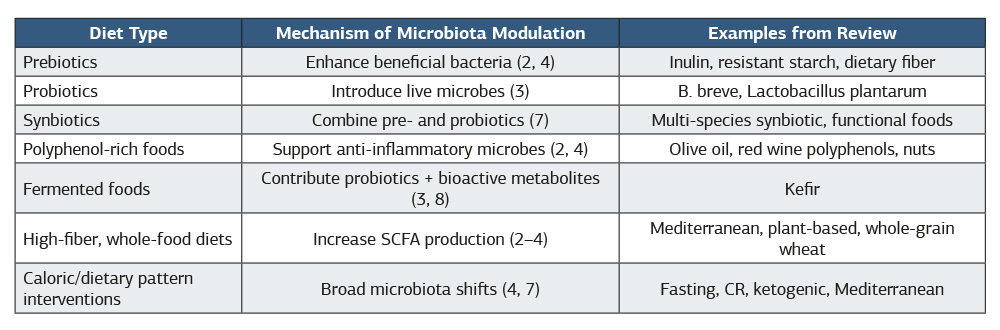
Data ItemsThe following variables were extracted from each included study: author(s), year, country, study design and population characteristics, type of dietary exposure (e.g., fiber, fat, specific diets), microbiota-related outcomes (e.g., diversity, SCFA production), metabolic health outcomes (e.g., BMI, insulin resistance, lipid profiles), and key findings along with reported study limitations. A summary of the 49 RCTs incorporated into this scoping review is presented in Table 1. These findings elucidate the complex relationship among diet, gut microbiota, and metabolic health. Studies were described according to their design, population, and dietary intervention, or relationship with the gut microbiota and metabolic parameters, and included key findings and limitations, with a synthesis of evidence across different dietary exposures and health outcomes.
Critical Appraisal of Individual Sources of EvidenceOnly RCTs were included in the scoping review, as stipulated at the outset through the inclusion criteria, to ensure methodological quality and uniformity of evidence sources. A formal critical appraisal or risk of bias assessment was not performed a priori, as per the adopted scoping review methodology. However, the RCT-only paradigm generates a basic level of trust in the review of evidence about diet and gut microbiota interactions and metabolic health.
Ethics ApprovalEthics committee approval was not required because this study was a scoping review based exclusively on previously published studies.
Statistical AnalysisNo quantitative meta-analysis or inferential statistical analysis was performed. Findings from the included randomized controlled trials were synthesized descriptively and presented narratively in accordance with scoping review methodology.
Reporting GuidelinesThe study was reported in accordance with the PRISMA-ScR guidelines.
Results
Selection of Sources of EvidenceA comprehensive literature search across multiple databases yielded a total of 2,615 records relevant to the relationship between diet, gut microbiota, and metabolic health outcomes. These records were imported into Rayyan, a systematic review management tool, where both automated and manual processes were conducted to remove duplicates. This step identified and removed 1,065 duplicate entries: 503 were resolved, 21 were not duplicates, and 541 were duplicates and deleted, leaving 2,704 unique records for screening.
The screening process involved a two-stage approach. First, titles and abstracts of the 2,074 records were reviewed independently by two reviewers. Based on inclusion criteria focusing on RCTs involving human participants and reporting on diet, microbiota, and metabolic outcomes, 671 studies were deemed potentially eligible. These were then subjected to full-text review.
Out of the 671 full-text articles assessed for eligibility, 54 articles could not be retrieved as they were not completed. Of the 49 RCTs that met all inclusion criteria, 49 were included in the final synthesis. The primary reasons for excluding the remaining 568 articles were that the study design was not an RCT, focused on non-human or pediatric populations, lacked microbiota or metabolic outcome data, or did not involve a clear dietary intervention.
The entire selection process is represented in the PRISMA-ScR flow diagram (Figure 1).
Characteristics of Sources of EvidenceIn this synthesis, 49 RCTs were included. These studies were conducted in 26 different countries, confirming the global aspect and diversity of research investigating the relationship between diet, microbiota, and metabolic health. Trials varied in length, with most spanning 4 to 24 weeks. Precise average times were poorly documented in the studies.
The majority of the studies employed valid methodological designs, primarily double-blind, placebo-controlled trials (e.g., a 12-week intervention among 53 overweight or obese adults), implying strict internal validity to reduce bias. The dietary interventions were heterogeneous, involving various functional foods, prebiotics, fibers, and fermented products. The three most prevalent interventions were (1) Polyphenol supplementation (e.g., 1.5 g/day Camu-camu) compared with placebo; (2) high-fiber cereal interventions, for example, rye products with 30g/day fiber vs refined wheat; (3) synergistic effect of oil and fiber supplementation, including Camelina sativa oil and resistant dextrin.
These interventions were assessed for their impact on gut microbial composition i.e., Bifidobacterium, Faecalibacterium and central markers of metabolic health such as glycemic control, lipid profile, inflammation, and body composition.
Collectively, this is a portfolio of trials that offers a well-executed collection of evidence examining the trinity of diet, gut microbiota, and metabolic disease. Nevertheless, differences in the type of intervention, outcome measurements, and sample size warranted an agreement on methods to be used in future trials. The 49 selected RCTs, along with their key features, are summarized in Supplementary Table 1, 2, 3 and 4. Each record describes the lead author, year, country, design, population, diet, microbiota modifications, metabolic health effects, main results, and limitations.
The included studies employed diverse dietary strategies, each targeting the gut microbiota through distinct mechanisms (Table 1). These interventions ranged from isolated prebiotic compounds to complex dietary patterns, with varying effects on microbial diversity, metabolite production, and downstream metabolic outcomes.
Critical Appraisal within Sources of EvidenceConsistent with the established scoping review methodology, a formal critical appraisal of the sources of evidence was not conducted.5 The procedure aligns with the purpose of a scoping review, which is to identify the extent and nature of the research rather than to assess the risk of bias or methodological quality.
However, all studies in this review were RCTs, which were defined a priori at the most stringent level of acceptable evidence in the inclusion criteria. By limiting inclusion to RCTs only, the review maintains a consistent threshold for methodological quality across sources.
This criterion enhances the internal validity and robustness of conclusions drawn from the pooled estimates while also allowing for the breadth of study populations, intervention types,, and outcomes measured, which is characteristic of the diet-microbiota-metabolism field.
Results of Individual Sources of EvidenceThe results of these 49 included RCTs were thematically grouped under three thematic domains that aligned with the scoping review objective: (1) the effect of diet on gut microbiota composition, (2) the relationship between gut microbiota and metabolic health outcome, and (3) approach to intervention that targets both domains simultaneously.
Diet and Gut MicrobiotaA variety of dietary interventions demonstrated clear effects on gut microbial profiles. For instance, Rustanti et al.7 documented a remarkably higher abundance of Lactobacillus plantarum after probiotic administration in women with T2D and downstream effects on the relative abundance of other taxa, such as Faecalibacterium. Salazar et al.7 reported that inulin-type fructans promoted the growth of particular Bifidobacterium spp. and inversely correlated with the concentration of SCFAs in feces. Iversen et al.8 found that a rye diet resulted in an increased abundance of butyrate-producing taxa, such as Agathobacter. On the other hand, a polyphenol- and fiber-fortified functional beverage has been shown to increase microbial diversity, although no modification in alpha diversity indexes was observed.9 These examples illustrate that a myriad of dietary inputs can differentially impact the microbial ecology, frequently favoring the growth of SCFA producers or barrier-supporting bacteria. Figure 2 is a graphical overview of the systemic effects of obese-type gut microbiota and microbial interactions with host physiology. The illustration is provided to visually integrate how obese-type gut microbiota efforts impact various metabolic organs and pathways together (e.g., host appetite, inflammation, and lipid and glucose metabolism).
Microbiota and Metabolic DiseasesMicrobial changes that correlated with beneficial changes in host metabolic markers were also observed in multiple studies. Kanazawa et al.10 recently reported that the use of a synbiotic was associated with increased levels of Bifidobacterium adolescentis and short-chain fatty acids (SCFAs), with a concurrent decrease in insulin resistance in obese adults with T2D. Medina-Vera et al.11 found that endotoxemia and inflammation were reduced after a functional food intervention in T2D patients , and this reduction correlated with higher levels of Akkermansia muciniphila. Horvath et al.12 also associated higher levels of Faecalibacterium prausnitzii with lower IL-6 and better lipid profiles, thereby strengthening the relationship between gut microbiota and systemic metabolic regulation.
Triangular Studies: Diet ➝ Microbiota ➝ Metabolic Improvement
Several studies presented evidence along the complete diet--microbiota--metabolism axis. Gao et al.13 reported that a polyphenol-enriched Mediterranean diet modulates bile acid signaling and enhances Roseburia and lipid metabolism. Gómez-Pérez et al.14 reported that fiber-induced microbial richness was associated with lower hepatic fat and C-reactive protein (CRP) among patients with non-alcoholic fatty liver disease (NAFLD). In a cross-over trial, Capurso et al.15 reported that sourdough bread intake increased gastrointestinal symptoms, as well as an abundance of Bifidobacterium, but failed to affect systemic biomarkers. Together, these trials suggest that specific dietary alterations can reprogram the microbiota, resulting in corresponding improvements in clinical symptoms.
Synthesis of ResultsThe 49 RCTs included in this review provide invaluable insights into the connections between diet, gut microbiota, and metabolic health. The conclusion was comprised of three synthesized, interrelated themes that corresponded with the guiding questions of the review.
1. Diet as a Microbiota Modulator
The diversity of dietary practices was significantly associated with gut microbial similarity. Interventions high in fermentable carbohydrates, plant polyphenols, and unsaturated fats have been found to favor taxa that are SCFA producers and strengthen the barrier. Studies involving whole foods (including legumes, whole grains, and fermented foods) were more likely to increase microbial richness than those focusing on isolated nutrients. This shift highlights the role of the food matrix and synergy in shaping microbial communities. Responses were heterogeneous, but interventions promoting a greater diversity of gut bacteria were, in general, associated with more robust ecological shifts.
2. Microbiota as a Mediator of Metabolic Health
The gut microbiota is often associated with metabolic benefits, particularly in relation to glucose homeostasis, lipid metabolism, and peripheral inflammation. These effects were frequently associated with microbial metabolites, including short-chain fatty acids (SCFAs) and secondary bile acids, both of which interact with host receptors, impacting insulin sensitivity and inflammatory tone. The size of the effect depended on baseline microbiota composition and participant status, thus making the overall metabolic benefits of microbiota modulation not universally guaranteed and perhaps ultimately dependent on the microbiome characteristics of individuals or populations.
3. Integrated Triangular Mechanisms
Slightly more than a third of the studies were retrospective and considered the entire route from dietary intervention to microbiota modification to metabolic profile modification. This triangle offered the strongest support for causation, showing that dietary change can lead to gut microbiota reconstruction, the reconstruction of which could, in turn, affect metabolism. The magnitude and duration of these effects, however, were variable, with modest changes in clinical parameters in some cases despite marked changes in microbial composition.
Schematic Overview of the Triangular Interaction in Dietary-Microbiota-Metabolic InterrelationshipFigure 3 provides a summary of the triangular correlation, where various dietary types, including high-fiber, Mediterranean, and Western diets, affect gut microbiota ecology and metabolism, which subsequently impact key metabolic and glycemic outcome measures, such as insulin sensitivity, BMI, and hepatic fat levels.
Trends and LimitationsHeterogeneity in response, however, also illustrates the complexity of analyzing host-microbiome interactions within humans. Across the body of evidence, it is apparent that diversity consistently stands out as a marker of health and resilience. Primary limitations included the short duration of interventions, restricted geographical sampling, and the lack of mechanistic data to connect certain microbes with specific outcomes. However, these shortcomings do not detract from the consistent conclusion that the microbiome is a critical determinant that mediates dietary effects on chronic metabolic diseases.
Discussion
This scoping review sought to systematically map and synthesize RCT evidence investigating the impact of dietary interventions on gut microbiota composition and its impact on metabolic health outcomes. By limiting inclusion to RCTs in human adults, the review is biased toward a high level of internal validity and methodological quality, thereby enabling stronger inferences about potential causal relationships. This finding was consistent with the primary purpose of elucidating how individual dietary components affect the microbiome and, in turn, influence host metabolism.5
The RCTs included provide consistent support for three fundamental conceptual themes that frame our synthesis. Diet as a driver of gut microbiota modulation was found in almost all included studies. Nutritional interventions with fermentable fibers, resistant starches, polyphenol-rich plant foods, and fermented dairy products have been demonstrated to modulate the microbial community structure, thereby increasing the diversity and abundance of health-associated taxa. Notably, taxa such as Bifidobacterium, Faecalibacterium prausnitzii, and Akkermansia muciniphila were commonly enriched after these interventions, echoing previous observations on the association of this microbiota with a favorable metabolic profile.2,3
Moreover, the role of the gut microbiota as a mediator of metabolic end-points was robustly supported by trials that correlated changes in the microbiota with changes in insulin sensitivity, blood lipids, circulating levels of inflammatory markers e.g., C-reactive protein (CRP), and hepatic fat content. Most of these effects were attributed to microbial metabolites, including short-chain fatty acids (SCFAs) and secondary bile acids, which act on host receptors to regulate glucose and lipid homeostasis.4
Although the studies were part of a global collaborative research project, low-income and middle-income countries were still underrepresented, contributing to cultural and dietary homogeneity in the evidence. The underrepresentation limits inferences to other dietary customs and microbiomes adapted to different ecological pressures.
In sum, the implications of the results are general. It can be suggested that targeted dietary recommendations modulating the microbiota can be considered an adjuvant for metabolic disease prevention and treatment. Future studies will need to prioritize specific microbial players and bioactive metabolites, particularly SCFAs, as therapeutic targets. Lastly, public health advocates can utilize this burgeoning evidence to lobby for microbiome-informed dietary guidelines aimed at enhancing both gut and metabolic health at a population level.2,4
Limitations
Considering the discussed limitations of the reviewed evidence, the field could benefit from a new era of longer-term RCTs that incorporate multi-omics profiling to more accurately define microbe–host metabolic interactions. The standardization of microbiota procedures, SCFA measurements, and the measurement of metabolic outcomes will enhance the comparability between studies. Finally, a wider representation of different geographical and cultural diets is necessary to extend the generalizability of results and enhance the understanding of microbiome plasticity across diverse environmental and genetic backgrounds.
Taken together, the review revealed the power of dietary interventions to program gut ecology in a way that promotes metabolic health, as well as provide a roadmap for more rigorous and inclusive explorations of the microbiome.
Conclusion
The findings of this scoping review offer compelling support for the central hypothesis that dietary interventions can meaningfully alter the gut microbiome, with subsequent effects on metabolic health. It was most evident in studies where these three components of the diet ‒microbiota‒metabolism interconnection were investigated. Together, the evidence suggests that gut microbiota modulation by crop-specific nutrition could be a promising approach for enhancing insulin sensitivity, lipid profiles, inflammatory markers, and other metabolic responses. Consistently observed changes (increases in SCFAs and decreases in C-reactive protein and pro-inflammatory cytokines) delineate putative causal pathways, but these need further mechanistic and longitudinal validation.
Furthermore, from a translational perspective, it appears that the growing body of microbiome-related diet research may increasingly support “personalized nutrition,” which integrates microbial profiling with individualized dietary advice. These strategies are particularly promising in the prevention and treatment of metabolic diseases, including T2D, obesity, and non-alcoholic fatty liver disease (NAFLD). These findings also support the use of microbiome-aware frameworks in public health nutrition policy, including incentives for high-fiber, plant-based, and fermented food patterns that have been shown to enhance beneficial microbial taxa.
Considering the discussed limitations of the reviewed evidence, the field could benefit from a new era of longer-term RCTs that incorporate multi-omics profiling to more accurately define microbe–host metabolic interactions. The standardization of microbiota procedures, SCFA measurements, and the measurement of metabolic outcomes will enhance the comparability between studies. Finally, a wider representation of different geographical and cultural diets is necessary to extend the generalizability of results and enhance the understanding of microbiome plasticity across diverse environmental and genetic backgrounds.
Taken together, the review revealed the power of dietary interventions to program gut ecology in a way that promotes metabolic health, as well as provide a roadmap for more rigorous and inclusive explorations of the microbiome.
Declarations
Ethics Declarations
The authors declare that all procedures performed in this study were conducted in accordance with institutional, national, and international ethical standards.
Animal and Human Rights Statement
This article is a scoping review based on previously published human studies. No new human or animal experiments were performed by the author.
Informed Consent
Informed consent was not required because this study used previously published data.
Data Availability
All data analyzed in this study were obtained from previously published articles cited in the reference list.
Conflict of Interest
The author declares no conflict of interest.
Funding
This research was funded by the Deanship of Scientific Research at Northern Border University, Arar, Saudi Arabia, under project number NBU-FFR-2025-2975-10.
Author Contributions (CRediT Taxonomy)
Conceptualization: N.S.A.
Methodology: N.S.A.
Investigation: N.S.A.
Data curation: N.S.A.
Formal analysis: N.S.A.
Writing – original draft: N.S.A.
Writing – review & editing: N.S.A.
Supervision: N.S.A.
Scientific Responsibility Statement
The author is responsible for the study conception and design, literature review, data interpretation, manuscript preparation, and approval of the final version of the manuscript.
Abbreviations
BMI: Body mass index
CRP: C-reactive protein
MetS: Metabolic syndrome
NAFLD: Non-alcoholic fatty liver disease
PRISMA-ScR: Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews
RCT: Randomized controlled trial
SCFA: Short-chain fatty acid
T2D: Type 2 diabetes
References
-
Thursby E, Juge N. Introduction to the human gut microbiota. Biochem J. 2017;474(11):1823-1836. doi:10.1042/bcj20160510
-
Valdes AM, Walter J, Segal E, Spector TD. Role of the gut microbiota in nutrition and health. BMJ. 2018;361:k2179. doi:10.1136/bmj.k2179
-
David LA, Maurice CF, Carmody RN, et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014;505(7484):559-563. doi:10.1038/nature12820
-
Tilg H, Moschen AR. Microbiota and diabetes: an evolving relationship. Gut. 2014;63(9):1513-1521. doi:10.1136/gutjnl-2014-306928
-
Arksey H, O’Malley L. Scoping studies: toward a methodological framework. Int J Soc Res Methodol. 2005;8(1):19-32. doi:10.1080/1364557032000119616
-
Peters MDJ, Marnie C, Tricco AC, et al. Updated methodological guidance for the conduct of scoping reviews. JBI Evid Synth. 2020;18(10):2119-2126. doi:10.11124/jbies-20-00167
-
Rustanti N, Murdiati A, Juffrie M, et al. Effect of probiotic Lactobacillus plantarum Dad-13 on metabolic profiles and gut microbiota in type 2 diabetic women: a randomized double-blind controlled trial. Microorganisms. 2022;10(9):1806. doi:10.3390/microorganisms10091806
-
Iversen KN, Dicksved J, Zoki C, et al. The effects of high fiber rye, compared to refined wheat, on gut microbiota composition, plasma short-chain fatty acids, and implications for weight loss and metabolic risk factors (the RyeWeight study). Nutrients. 2022;14(8):1669. doi:10.3390/nu14081669
-
Santamarina A, Filho V, de Freitas J, et al. Nutraceutical blends promote weight loss, inflammation reduction, and better sleep: the role of Faecalibacterium prausnitzii in overweight adults—a double-blind trial. Mol Nutr Food Res. 2025:e202400806.
-
Kanazawa A, Aida M, Yoshida Y, et al. Effects of synbiotic supplementation on chronic inflammation and the gut microbiota in obese patients with type 2 diabetes mellitus: a randomized controlled study. Nutrients. 2021;13(2):558. doi:10.3390/nu13020558
-
Medina-Vera I, Sanchez-Tapia M, Noriega-López L, et al. A dietary intervention with functional foods reduces metabolic endotoxaemia and attenuates biochemical abnormalities by modifying faecal microbiota in people with type 2 diabetes. Diabetes Metab. 2019;45(2):122-131. doi:10.1016/j.diabet.2018.09.004
-
Horvath A, Leber B, Feldbacher N, et al. Effects of a multi-species synbiotic on glucose metabolism, lipid marker, gut microbiome composition, gut permeability, and quality of life in diabesity: a randomized, double-blind, placebo-controlled pilot study. Eur J Nutr. 2020;59(7):2969-2983. doi:10.1007/s00394-019-02135-w
-
Gao P, Rinott E, Dong D, et al. Gut microbial metabolism of bile acids modifies the effect of Mediterranean diet interventions on cardiometabolic risk in a randomized controlled trial. Gut Microbes. 2024;16(1):2426610. doi:10.1080/19490976.2024.2426610
-
Gomez-Perez A, Ruiz-Limon P, Salas-Salvado J, et al. Gut microbiota in non-alcoholic fatty liver disease: a PREDIMED-Plus trial subanalysis. Gut Microbes. 2023;15(1):2223339.
-
Capurso G, Tacelli M, Vanella G, et al. Managing complications of chronic pancreatitis: a guide for the gastroenterologist. Expert Rev Gastroenterol Hepatol. 2023;17(12):1267-1283. doi:10.1080/17474124.2023.2295498
Figures
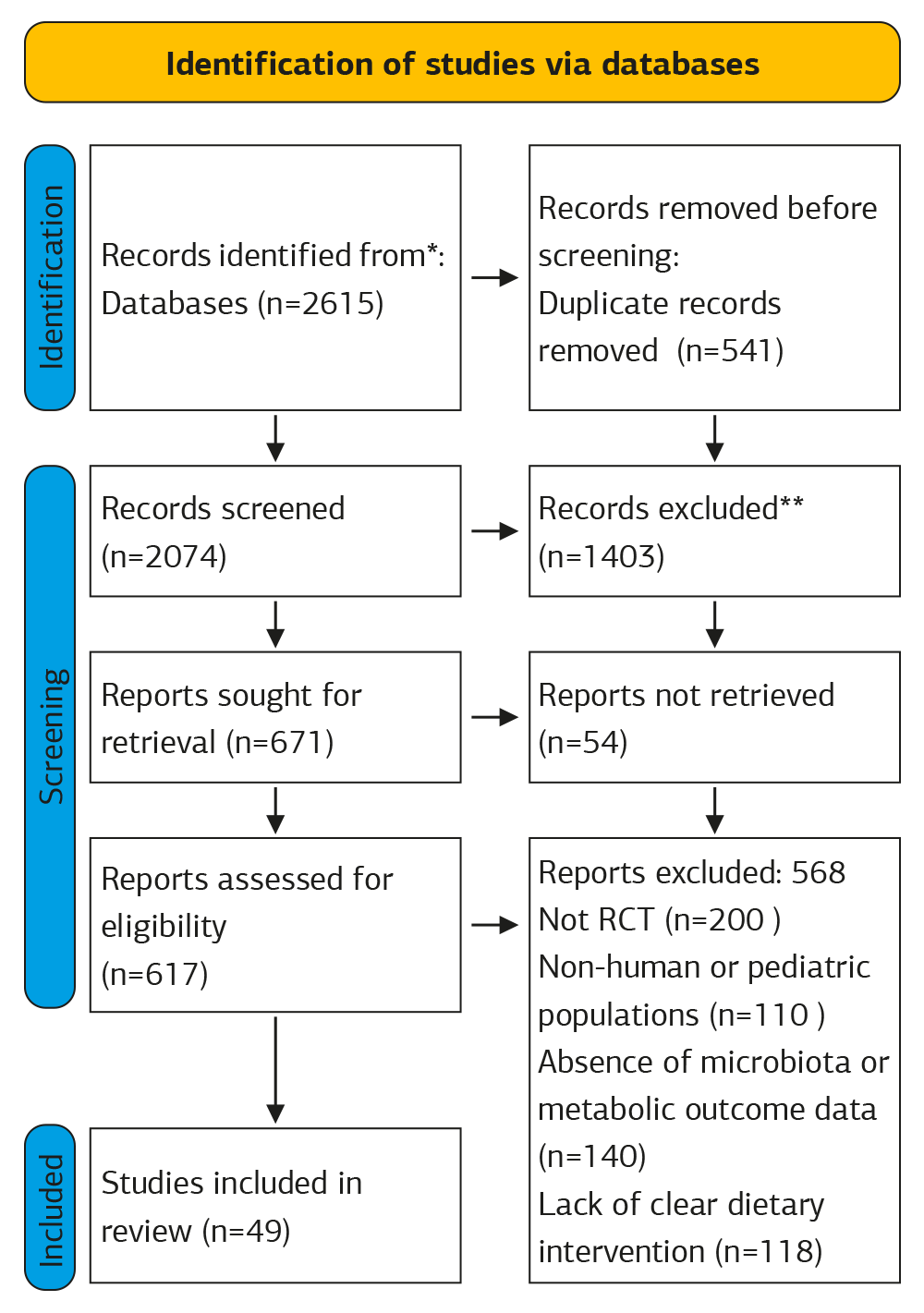
Figure 1. PRISMA-ScR flow diagram of the study selection process
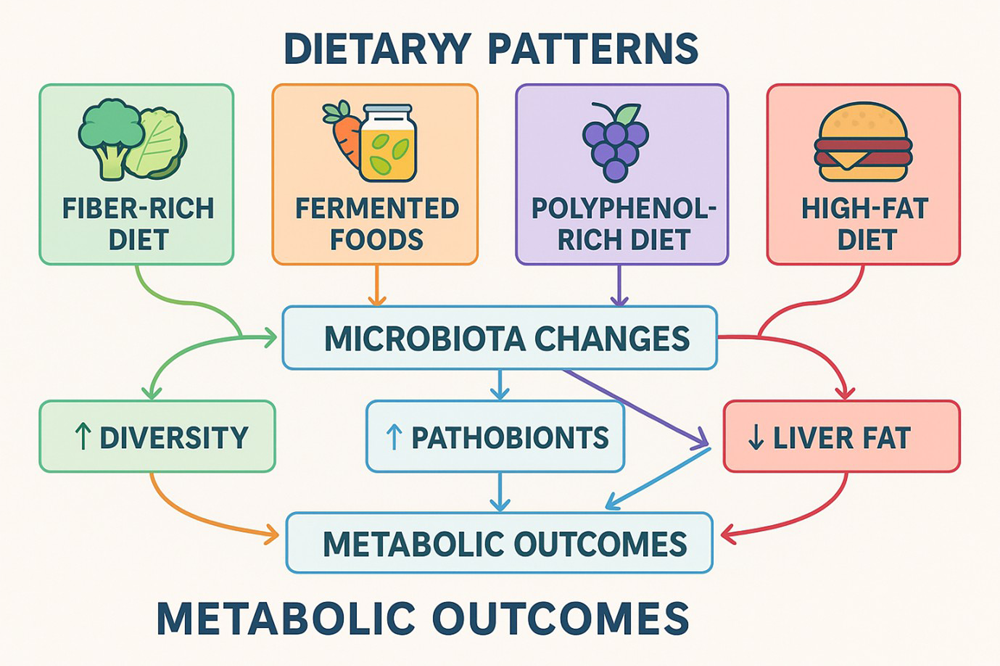
Figure 2. Schematic illustration showing the multi-organ effects of an obese-type gut microbiota, including impacts on satiety, inflammation, and metabolic regulation.
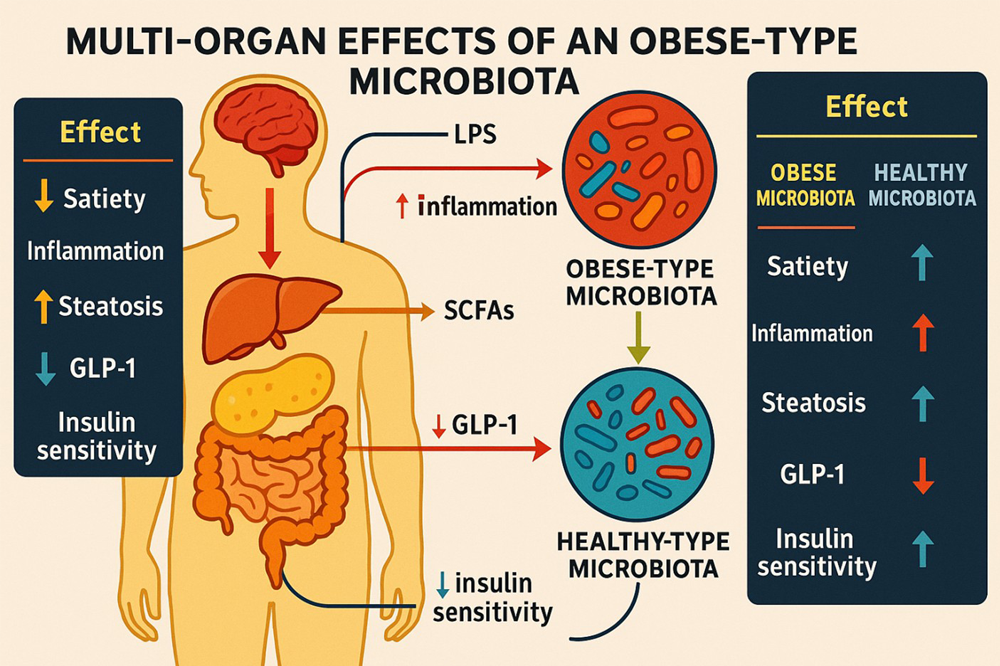
Figure 3. Conceptual summary diagram illustrating how various dietary patterns (e.g., high fiber, Mediterranean diet, Western diet) influence gut microbiota composition and subsequently affect metabolic outcomes such as insulin sensitivity, BMI, and liver fat.
Tables
Table 1. Summary of dietary intervention types, their proposed mechanisms of microbiota modulation, and representative examples from included RCTs
Note: Citations reflect evidence supporting the mechanisms identified in this scoping review
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Nasser Salem Alqahtani. Gut microbiota, diet, and metabolic diseases: a triangular relationship. Ann Clin Anal Med 2026;17(6):648-654. doi:10.4328/ACAM.22811
- Received:
- July 13, 2025
- Accepted:
- August 25, 2025
- Published Online:
- September 22, 2025
- Printed:
- June 1, 2026

