Metabolic syndrome’s echo on the obese heart: a closer look at diastolic function and myocardial performance index
Metabolic syndrome and obese heart
- Received:
- August 6, 2025
- Accepted:
- September 29, 2025
- Published Online:
- September 30, 2025
- Printed:
- October 1, 2025
Authors
Abstract
AimThis study investigated the effect of the presence or absence of metabolic syndrome (MetS) on cardiac structure and function in obese individuals.
MethodsA retrospective, cross-sectional, and observational study design was used. 125 patients diagnosed with obesity were included in the study and divided into two groups according to the presence of MetS. Demographic, clinical, and echocardiographic data of the patients were analyzed.
ResultsNo significant difference was found in diastolic functions and myocardial performance index (MPI) between obese individuals with and without MetS. However, a trend was observed in these parameters compared to the normal population in both groups. Systolic blood pressure, triglyceride, LDL-cholesterol, and total cholesterol levels were significantly higher in the MetS group than in the non-MetS group.
ConclusionObesity may have adverse effects on cardiac structure and function, regardless of the presence of MetS. Therefore, echocardiography is an important tool for the assessment and management of cardiovascular risks in obese individuals, even if they do not develop MetS. Targeted strategies such as lifestyle changes, pharmacological treatments, and regular cardiac monitoring should be developed to protect and improve the cardiovascular health of patients with obesity and MetS.
Keywords
Introduction
Obesity and metabolic syndrome (MetS) are two of the most significant public health challenges, with increasing global prevalence and well-documented associations with cardiovascular (CV) diseases 1. Obesity is a multifactorial condition characterized by excessive body weight, systemic inflammation, oxidative stress, and metabolic disturbances, whereas MetS represents a cluster of CV risk factors, including central obesity, insulin resistance, dyslipidemia, and hypertension 2. The combined effects of these conditions promote adverse structural and functional changes in the myocardium, leading to a markedly increased risk of CV morbidity and mortality 3. Given this burden, early and accurate assessment of myocardial function is essential for effective prevention and management strategies. While numerous studies have shown that both obesity and MetS impair myocardial function 4, direct comparisons between individuals with obesity alone and those with both obesity and MetS remain scarce, particularly regarding early markers of dysfunction.
Echocardiography is a non-invasive and widely available modality for assessing myocardial performance. The myocardial performance index (MPI) is a simple, reproducible echocardiographic parameter that integrates systolic and diastolic function 5 and has proven valuable in identifying subclinical myocardial impairment 6. This study aims to compare MPI values between obese patients with and without MetS, providing deeper insight into the additive effects of MetS on myocardial function and reinforcing the utility of MPI in CV risk stratification and management in this high-risk group.
Materials and Methods
This retrospective, cross-sectional, single-center observational cohort study aims to quantitatively evaluate the synergistic effects of obesity and metabolic MetS comorbidity on left ventricular diastolic dysfunction (LVDD) and myocardial performance MPI. The STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines design the study protocol, and data collection and analysis processes were conducted per the principles of Good Clinical Practice 7.
The study population consisted of adults aged 18-75 years who were followed up at the Cardiology and Endocrinology Clinic of Bilecik Education and Research Hospital between 01.01.2022-31.12.2024, diagnosed with obesity with a BMI ≥30 kg/m2 according to the TEMD (Turkish Society of Endocrinology and Metabolism) criteria 8 and underwent echocardiographic examination. The patients’ demographic, medical history, anthropometric, and laboratory data were extracted from the hospital data system by a double-blind method (2 independent researchers). Inclusion criteria included a complete echocardiographic data set (LVEF, E/A ratio, MPI), lipid profile, fasting glucose measurement in the last 3 months, and the recording of anthropometric measurements by WHO standards. Patients will be divided into two groups according to the presence of metabolic syndrome according to NCEP- ATP III criteria: (1) Obese Patients with Metabolic Syndrome and (2) Obese Patients Without Metabolic Syndrome. Metabolic syndrome will be diagnosed by the presence of at least three of the following NCEP-ATP III criteria 9.
Metabolic syndrome criteria:•Abdominal Obesity (Waist circumference); Male ≥94 cm, female ≥80 cm
•Hypertriglyceridemia ≥150 mg/dL
•HDL-Cholesterol Male <40 mg/dL, female <50 mg/dL
•Blood Pressure Systolic ≥130 mmHg or diastolic ≥85 mmHg
•Fasting Glucose ≥100 mg/dL
Exclusion criteria:•Known cardiovascular disease history (Angiographically
confirmed CAD (≥50% stenosis), NYHA Class II-IV heart failure
•Moderate-severe valvular heart disease
•Renal Dysfunction (eGFR <60 mL/min/1.73m2 according to
CKD-EPI formula)
•Liver Disease (Child-Pugh Score ≥B8 or ALT >3xULN)
•Active infection or inflammatory disease
•During pregnancy or lactation
•Thyroid dysfunction
Echocardiographic ProtocolAll examinations were performed using the Philips EPIQ CVx device and the X5-1 matrix probe in accordance with the ASE (American Society of Echocardiography) guidelines 10.
E and A waves will be measured from mitral inflow using pulsed-wave Doppler, and the E/A ratio will be calculated to evaluate left ventricular diastolic function. In addition, E’ wave velocities measured from septal and lateral regions of the mitral annulus using tissue Doppler imaging (TDI) and E/e’ ratio were evaluated. Myocardial performance index (MPI) measurement was calculated according to the IVCT+IVRT/ET formula using isovolumetric contraction time (IVCT), isovolumetric relaxation time (IVRT), and ejection time (ET). (Figure 1). Each parameter was measured in 3 consecutive cardiac cycles, and their averages were taken. Images were archived in DICOM format and analyzed unthinkingly by two cardiologists with expert- level echocardiography certificates. In echocardiographic measurements, interobserver ICC was 0.92 (95% CI 0.88-0.95). Data extraction error rate was <0.5% (6 sigma quality control).
Statistical AnalysisTo determine the statistical significance of quantitative differences between experimental groups, a two-tailed unpaired t-test and one-way ANOVA with Tukey’s multiple comparison test (95% confidence level) were conducted using GraphPad 8.1 software (GraphPad Software, San Diego, California). On the other hand, parts of whole analyses were used to illuminate the relationships between categorical variables in qualitative data. Descriptive and fraction of total statistics were applied to reveal the basic characteristics of the obtained data (mean, standard deviation, median, percentage, etc.). The significance level was always set at a 95% confidence interval, denoted as *p≤0.05, p≤0.01, *p≤0.001, ****p≤0.0001. Non-significant p-values are presented without any asterisks. The results are presented as mean ± standard deviation (SD).
Ethical ApprovalThis study was approved by the Bilecik Şeyh Edebali University, Faculty of Medicine Ethics Committee (Date: 2024-10-02, No: 4).
Results
Participant Demographics and Clinical FeaturesThe study included 125 obese patients (74 females, 51 males): 73 (58.4%) had MetS and 52 (41.6%) did not. Mean age was similar between groups (MetS: 56.7 ± 9.2 vs. non-MetS: 54.9 ± 9.8 years, p=0.288). The female prevalence was higher in the MetS group (p < 0.05). Anthropometric measures showed no significant differences: BMI (31.5 ± 1.4 vs. 31.7 ± 2.5 kg/m2, p = 0.566) and waist circumference (102.8 ± 14.2 vs. 104.0 ± 16.1 cm, p = 0.664). Cardiometabolic parameters revealed higher systolic BP (137.5 ± 62.1 vs. 125.9 ± 32.6 mmHg, p=0.005) and triglycerides (185.1 ± 64.0 vs. 135.3 ± 26.2 mg/dL, p<0.001) in the MetS group. Diastolic BP was borderline significant (84.5 ± 10.3 vs. 80.7 ± 11.7 mmHg, p=0.055). LDL (137.5 ± 62.1 vs. 111.0 ± 48.8 mg/dL, p = 0.012) and total cholesterol (218.4 ± 58.3 vs. 183.5 ± 49.0 mg/dL, p = 0.001) levels were significantly higher in patients with MetS. Fasting glucose (132.5 ± 26.7 vs. 125.9 ± 32.6 mg/dL, p=0.219) and HDL (43.9 ± 8.7 vs. 45.2 ± 4.8 mg/dL, p=0.315) showed no significant difference.
Echocardiographic FindingsDiastolic function parameters showed no statistically significant differences between the groups. Mitral E velocity was slightly lower in the MetS group compared to the non- MetS group (64.4 ± 12.8 vs. 67.5 ± 10.4 cm/s, p=0.156), while mitral A velocity was similar (74.5 ± 18.5 vs. 73.7 ± 9.5 cm/s, p = 0.792). Lateral mitral annulus early diastolic velocity (Em) measured by tissue Doppler imaging was lower in the MetS group (13.2 ± 2.5 vs. 14.2 ± 2.1 cm/s), though this difference did not reach significance (p = 0.254). Diastolic indices, such as the E/A ratio (0.89 ± 0.21 vs. 0.92 ± 0.15, p = 0.459) and E/Em ratio (5.11 ± 1.68 vs. 4.90 ± 1.24, p = 0.454), were comparable between groups. Systolic time intervals, including isovolumetric contraction time (IVCT: 60.5 ± 12.9 vs. 62.2 ± 12.8 ms, p = 0.476), isovolumetric relaxation time (IVRT: 99.0 ± 19.3 vs. 97.2 ± 16.5 ms, p = 0.589), and ejection time (ET: 279.4 ± 40.2 vs. 281.4 ± 31.0 ms, p = 0.758), also showed no significant differences. Both groups had similar MPI values (0.58 ± 0.11 vs. 0.57 ± 0.10, p = 0.694). Full details of group-wise echocardiographic parameters are summarized in Table 1.
Correlation analysis revealed a positive association of MPI with systolic blood pressure (r = 0.28, p = 0.006), triglycerides (r = 0.33, p < 0.001), and fasting glucose (r = 0.21, p = 0.022). HDL levels positively correlated with E/A ratio (r = 0.25, p = 0.008), while no other significant correlations were found (Table 2). A gender-based analysis revealed a higher BMI in obese women with MetS compared to women without MetS (32.23 ± 2.83 vs. 31.54 ± 1.67 kg/m2, p = 0.022). Total cholesterol (p=0.007) and triglycerides (p < 0.001) were significantly elevated in both men and women with MetS versus controls. In women, systolic BP (138.2 ± 10.2 vs. 127.0 ± 14.6 mmHg, p = 0.004) and fasting glucose (133.4 ± 27.5 vs. 115.1 ± 29.5 mg/dL, p = 0.031) were significantly higher in the MetS group, while no such differences were observed in men. No significant differences were found between genders in echocardiographic parameters, inflammatory markers, or diastolic indices (p>0.05). Full gender- based distributions are presented in Table 3. Figure 2 and Figure 3 show the comparison of demographic, anthropometric, and echocardiographic measurements between the groups.
Discussion
The main finding of this study is that obesity is associated with early cardiac dysfunction, independent of the presence of metabolic syndrome (MetS). Our findings demonstrate that obesity itself contributes to adverse structural and functional cardiac changes, supporting its recognition as an independent cardiovascular (CV) risk marker. These results emphasize the importance of evaluating obesity’s direct effects on cardiac morbidity, apart from metabolic comorbidities. Even in obese individuals without MetS, a trend toward impaired diastolic function, reflected by reduced E/A ratio, decreased E/Em, and elevated myocardial performance index (MPI), was observed. These findings align with emerging evidence that “metabolically healthy obesity” is not truly benign, as subclinical myocardial changes may occur early in disease progression. A large-scale cohort study by Rozenbaum et al. (2019) similarly reported increased diastolic dysfunction risk in metabolically healthy obese individuals compared to normal-weight controls 11.
The lack of statistically significant differences in diastolic parameters between obese groups with and without MetS in our study suggests that obesity-related myocardial alterations may develop independently of the synergistic burden of MetS.
One explanation is that visceral adipose tissue (VAT) promotes systemic inflammation and lipotoxicity, leading to diastolic dysfunction 12. Proinflammatory cytokines such as IL-6 and TNF-α, as well as adipokines like leptin, are known to impair endothelial function, promote fibrosis, and induce myocyte hypertrophy 13. A recent study demonstrated a direct, MetS-independent association between VAT volume and left ventricular diastolic dysfunction 14. Additionally, free fatty acids (FFA), key mediators of myocardial lipotoxicity, contribute to oxidative stress and impaired calcium ATPase pump activity, delaying calcium reuptake and diastolic relaxation 15. These mechanisms are consistent with the prolonged isovolumetric relaxation time (IVRT) and reduced diastolic filling observed in both obese groups in this study.
The strong correlations between MPI and factors such as systolic blood pressure (SBP), triglycerides, and fasting glucose further underscore the interplay between hemodynamic and metabolic stress in myocardial remodeling. Increased left ventricular ejection fraction (LVEF), often observed in obesity, may initially reflect compensatory adaptation but is associated with elevated wall stress and fibrosis, as reported in earlier studies linking increased LVEF to higher MPI 16. Hypertriglyceridemia, another hallmark of obesity, has been shown to disrupt myocardial energy metabolism through VLDL accumulation, resulting in lipotoxic damage and diastolic dysfunction 17. Our results support these findings, showing a strong association between triglyceride levels and MPI.
Gender-specific analysis in this study revealed that obese women with MetS exhibited more pronounced increases in BMI, SBP, and fasting glucose compared to men, reflecting gender- related pathophysiological differences. Estrogen decline in postmenopausal women exacerbates visceral fat deposition, insulin resistance, and myocardial fibrosis 18. Moreover, leptin resistance, more common in women, may heighten sympathetic nervous system activation, increasing hypertension risk and myocardial ischemia 19,20. These results emphasize the need for sex-specific approaches to CV risk stratification and treatment 21.
Overall, our findings highlight the clinical relevance of echocardiographic screening in obese patients, regardless of MetS status. Subclinical changes in MPI and diastolic function parameters can serve as early warning signs of cardiac dysfunction and may help guide preventive interventions 22. Identifying early myocardial impairment in obesity could lead to timely lifestyle modification, medical management, and closer follow-up, potentially mitigating long-term cardiovascular complications.
Limitations
The limitations of this study include its retrospective, single- center design and the lack of measurements such as epicardial adipose tissue (EAT) thickness. Although causality cannot be established, the findings remain clinically relevant. A recent study identified EAT thickness ≥7 mm as an independent predictor of diastolic dysfunction 23, highlighting the value of incorporating this parameter in future research. Additionally, the homogeneity and regional selection of the study population limit generalizability, but also guide future work. Prospective studies and multi-omics approaches may better define the causal pathways linking obesity to cardiac dysfunction.
Conclusion
In conclusion, this study shows that obesity has a central role in cardiovascular risk assessment and may trigger myocardial dysfunction independently of MS. Early detection of subclinical findings has the potential to reduce cardiac morbidity through lifestyle interventions (diet, exercise) and targeted pharmacological treatments. Clinicians should consider routine echocardiographic evaluation in obese individuals and develop personalized treatment strategies. Furthermore, recognizing obesity as a disease and managing it with multidisciplinary approaches may improve cardiovascular survival.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of Bilecik Şeyh Edebali University, Faculty of Medicine (Date: 2024-10-02, No: 4)
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
References
-
Hajek A, Kretzler B, König HH. Prevalence and correlates of obesity among the oldest old. A systematic review, meta-analysis and meta-regression. Geriatr Gerontol Int. 2022;22(5):373-83.
-
Silveira Rossi JL, Barbalho SM, Reverete de Araujo R, Bechara MD, Sloan KP, Sloan LA. Metabolic syndrome and cardiovascular diseases: going beyond traditional risk factors. Diabetes Metab Res Rev. 2022;38(3):e3502.
-
Rus M, Crisan S, Andronie-Cioara FL, et al. Prevalence and risk factors of metabolic syndrome: a prospective study on cardiovascular health. Medicina (Kaunas). 2023;25;59(10):1711.
-
Preda A, Carbone F, Tirandi A, Montecucco F, Liberale L. Obesity phenotypes and cardiovascular risk: from pathophysiology to clinical management. Rev Endocr Metab Disord. 2023;24(5):901-19.
-
Fernandes JMG, de Oliveira Romão B, Rivera IR, et al. Clinical value of myocardial performance index in patients with isolated diastolic dysfunction. Cardiovasc Ultrasound. 2019;17(1):17.
-
Kılıç R, Aslan M, Nas N, Güzel T. Relationship between presystolic wave and subclinical left ventricular dysfunction as assessed by myocardial performance index in patients with metabolic syndrome. Int J Cardiovasc Imaging. 2023;39(11):2175-82.
-
von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370(9596):1453-7.
-
T Sabuncu, S Kıyıcı, MA Eren, et al. Summary of Bariatric surgery guideline of the Society of Endocrinology and Metabolism of Turkey. Turk J Endocrinol Metab. 2017;21(4):140-7.
-
National Cholesterol Education Program (NCEP) Expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) final report. Circulation. 2002;106(25):3143-421.
-
Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28(1):1-39.e14.
-
Rozenbaum Z, Topilsky Y, Khoury S, Pereg D, Laufer-Perl M. Association of body mass index and diastolic function in metabolically healthy obese with preserved ejection fraction. Int J Cardiol. 2019;277:147-52.
-
Huang CC, Yeh HY, Lin R. et al. Inhibition of visceral adipose tissue-derived pathogenic signals by activation of adenosine A2AR improves hepatic and cardiac dysfunction of NASH mice. Am J Physiol Gastrointest Liver Physiol. 2024;326(4):G385-G397.
-
Zhazykbayeva S, Hassoun R, Herwig M, et al. Oxidative stress and inflammation distinctly drive molecular mechanisms of diastolic dysfunction and remodeling in female and male heart failure with preserved ejection fraction rats. Front Cardiovasc Med. 2023;10:1157398.
-
Qu Y, Liu J, Li J, et al. Association of abdominal adiposity, hepatic shear stiffness with subclinical left-ventricular remodeling evaluated by magnetic resonance in adults free of overt cardiovascular diseases: a prospective study. Cardiovasc Diabetol. 2023;22(1):99.
-
Thirumathyam R, Richter EA, Goetze JP, et al. Investigating the roles of hyperglycaemia, hyperinsulinaemia and elevated free fatty acids in cardiac function in patients with type 2 diabetes via treatment with insulin compared with empagliflozin: protocol for the HyperCarD2 randomised, crossover trial. BMJ Open. 2022;12(8):e054100.
-
Tadic M, Cuspidi C, Pencic B, et al. Association between myocardial work and functional capacity in patients with arterial hypertension: an echocardiographic study. Blood Press. 2021;30(3):188-95.
-
Jiang L, Xu HY, Li Y, et al. The differential effects of dyslipidemia status and triglyceride-glucose index on left ventricular global function and myocardial microcirculation in diabetic individuals: a cardiac magnetic resonance study. Cardiovasc Diabetol. 2024;23(1):345.
-
Sivasinprasasn S, Sa-Nguanmoo P, Pratchayasakul W, Kumfu S, Chattipakorn SC, Chattipakorn N. Obese-insulin resistance accelerates and aggravates cardiometabolic disorders and cardiac mitochondrial dysfunction in estrogen- deprived female rats. Age (Dordr). 2015;37(2):28.
-
Omoto ACM, do Carmo JM, Nelson B, et al. Central nervous system actions of leptin improve cardiac function after ischemia-reperfusion: Roles of sympathetic innervation and sex differences. J Am Heart Assoc. 2022;11(21):e027081.
-
Jones AA, Framnes-DeBoer SN, Shipp A, Arble DM. Caloric restriction prevents obesity- and intermittent hypoxia-induced cardiac remodeling in leptin-deficient ob/ob mice. Front Physiol. 2022;13:963762.
-
Ardahanlı İ, Özkan Hİ, Özel F, Gurbanov R, Teker HT, Ceylani T. Infrared spectrochemical findings on intermittent fasting-associated gross molecular modifications in rat myocardium. Biophys Chem. 2022;289:106873.
-
Akhan O, Ardahanli I. Hypoglycemia in the emergency, is there any effect on endothelial and diastolic functions? Echocardiography. 2021;38(3):450-9.
-
Schulz A, Backhaus SJ, Lange T, et al. Impact of epicardial adipose tissue on cardiac function and morphology in patients with diastolic dysfunction. ESC Heart Fail. 2024;11(4):2013-22.
Figures
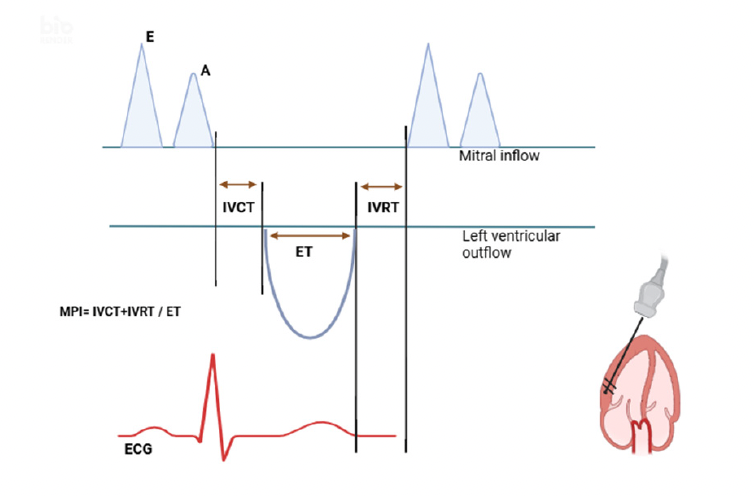
Figure 1. Myocardial performance index measurement
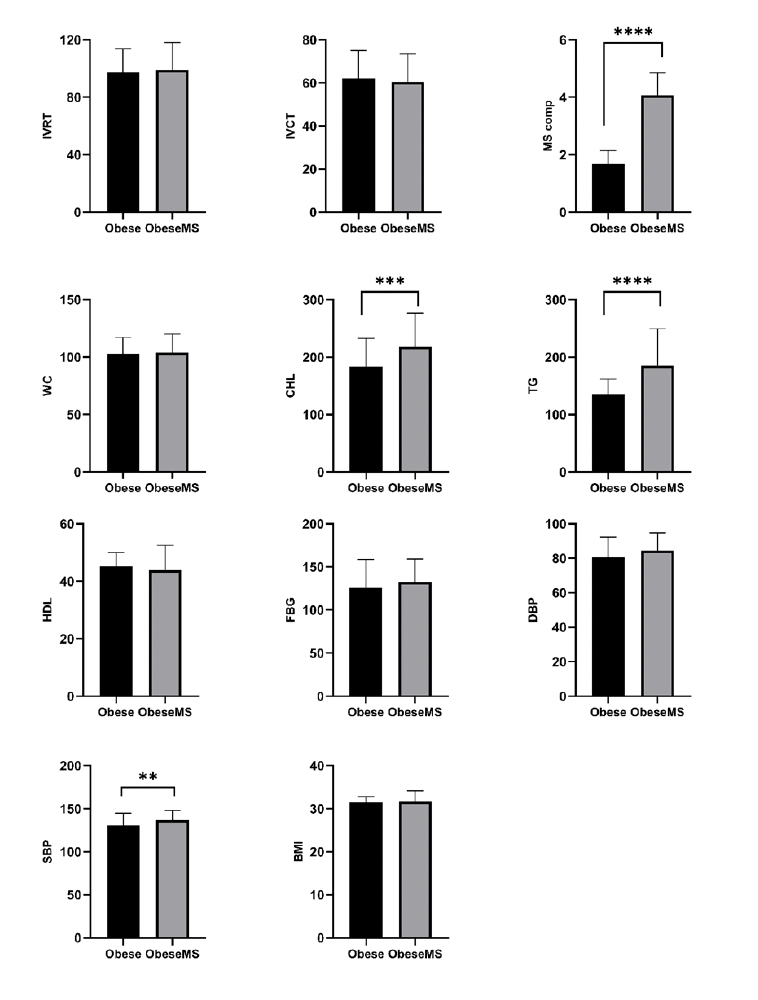
Figure 2. Comparison of demographic, anthropometric, and echocardiographic measurements between groups
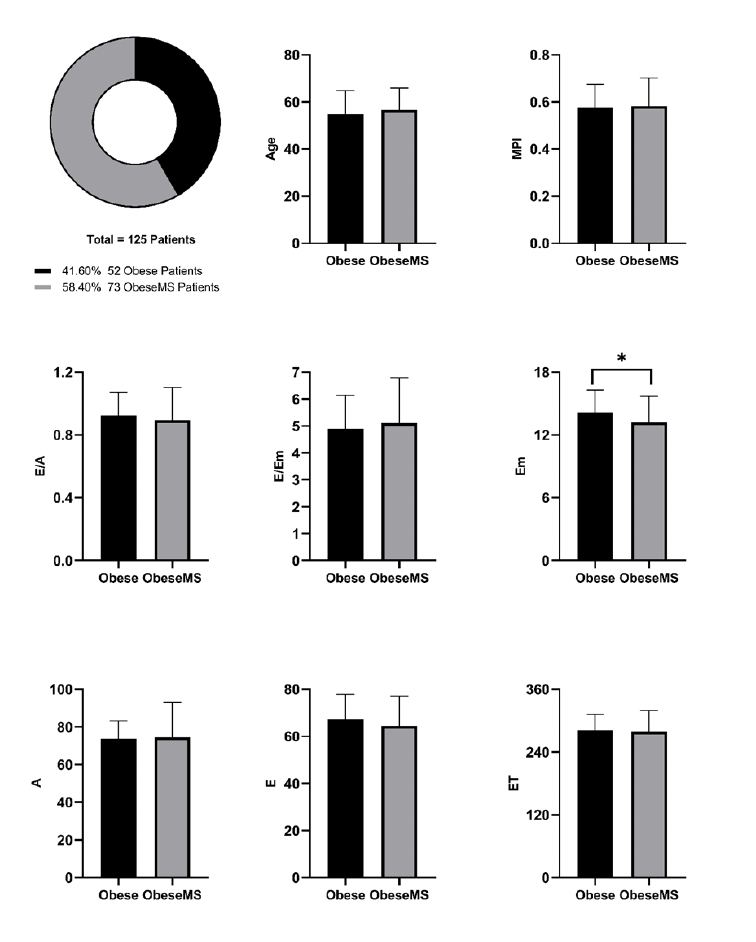
Figure 3. Comparison of demographic, anthropometric, and echocardiographic measurements between groups
Tables
Table 1. Distribution of patients with and without MetS

SBP = systolic blood pressure; DBP = diastolic blood Pressure; HDL-C = high dendsity lipoprotein cholesterol; LDL-C = low-density lipoprotein cholesterol; IVRT = isovolumetric relaxation time; IVCT = isovolumetric contraction time; MPI = myocardial performance index; MetS = metabolic syndrome
Table 2. Significant correlations between myocardial performance index and cardiometabolic parameters
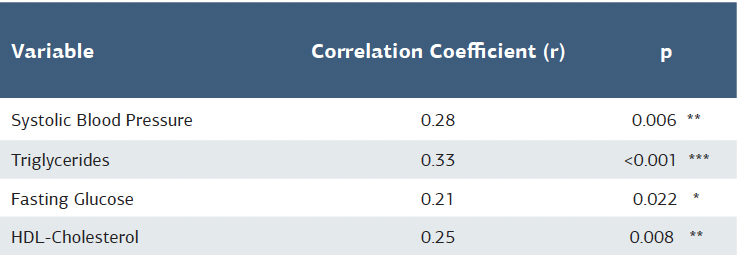
* p<0.05, ** p<0.01, *** p<0.001 (Pearson correlation analysis)
Table 3. Distribution of patients with and without MetS by gender
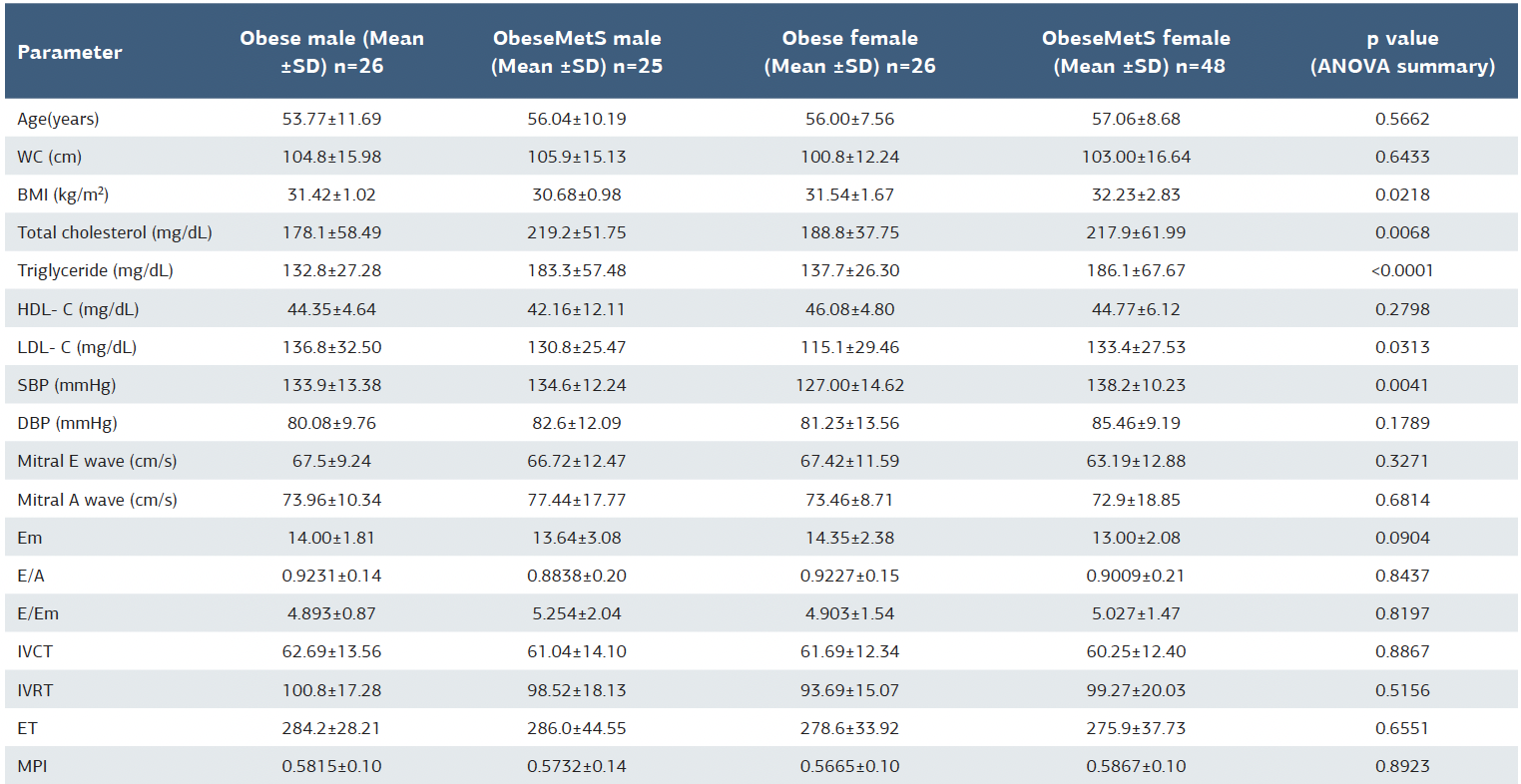
SBP = systolic blood pressure; DBP = diastolic blood Pressure; HDL-C = high-density lipoprotein cholesterol; LDL-C = low-density lipoprotein cholesterol; IVRT = isovolumetric relaxation time; IVCT = isovolumetric contraction time; MPI = myocardial performance index; MetS = metabolic syndrome; WC = waist circumference; BMI = body mass index
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Murat Özmen, Faik Özel, Ramazan Aslan, Erhan Arıkan, İsa Ardahanlı. Metabolic syndrome’s echo on the obese heart: A closer look at diastolic function and myocardial performance index. Ann Clin Anal Med 2025;16(10):741-746

