Can Dyslipidemia be a significant factor in myofascial pain syndrome?
Dyslipidemia and myofascial pain syndrome
- Received:
- August 19, 2025
- Accepted:
- September 22, 2025
- Published Online:
- September 30, 2025
- Printed:
- October 1, 2025
Authors
Abstract
AimTo investigate the presence of myofascial pain syndrome (MPS) in patients whose lipid profiles were assessed and to examine the relationship of lipid levels with pain and muscle spasm.
MethodsIn this study, 197 patients whose lipid profiles were evaluated were assessed for the presence of characteristic trigger points in the neck, back, and shoulder muscles, referred pain in a reference zone, and the state of muscle spasm.
ResultsThe groups were classified as dyslipidemic and healthy based on their lipid levels. The prevalence of MPS in the dyslipidemic group was found to be significantly higher compared to the group with normal lipid levels.
ConclusionA strong association was identified between dyslipidemia and MPS, and individuals with dyslipidemia were found to have a higher likelihood of developing MPS.
Keywords
Introduction
Myofascial pain syndrome (MPS) is a disorder characterized by the presence of palpable, tender, and active myofascial trigger points in the skeletal muscles 1. The etiology of the disease is not fully understood; however, factors such as trauma, excessive muscle strain, fatigue, stress, and structural and genetic abnormalities have been implicated 2. These trigger points are characteristically painful upon pressure and cause referred pain 3. Taut bands on palpation, local twitch responses, localized tingling sensations, jump signs, and referred pain are the most commonly observed findings in MPS 4. The prevalence of MPS in individuals aged 30–60 years has been reported as 37% in men and 65% in women 5.
In recent years, it has been proposed that the pathophysiology of MPS may not be limited to the musculoskeletal system alone and that systemic factors may also play a role. Furthermore, local biochemical microscopic analyses conducted on active myofascial trigger points have shown elevated levels of inflammatory mediators, neuropeptides, catecholamines, and pro-inflammatory cytokines compared to asymptomatic muscle tissue 6. In this context, there is growing research into the effects of lipid levels on MPS 7. Dyslipidemia is a clinical condition characterized by one or more abnormalities (either elevations or reductions) in total cholesterol, low-density lipoprotein (LDL), high-density lipoprotein (HDL), or triglyceride (TG) levels. It may have both genetic and environmental origins 8. According to the guidelines for the diagnosis and treatment of dyslipidemia, screening should include total cholesterol, TG, HDL cholesterol (HDL-c), LDL cholesterol (LDL-c), and non-HDL cholesterol. The Turkish Endocrinology and Metabolism Society defines the following as optimal values: TG < 150 mg/dL, total cholesterol < 200 mg/dL, LDL < 70 mg/dl, and HDL > 60 mg/ dL 9.
In the literature, some studies have reported a potential association between musculoskeletal symptoms and hyperlipidemia 10,11,12. These studies have emphasized that elevated lipoprotein and lipid levels in symptoms such as oligoarthritis, Achilles tendinitis, arthralgia in various joints, migratory polyarthritis, and myalgia may be related to a disorder in lipid metabolism 10,11. This relationship has also been noted in fibromyalgia syndrome (FMS), similarly to MPS 13. It has been highlighted that hyperlipidemia and other related cardiovascular comorbidities observed in patients with FMS may often be due to the strong association between FMS and obesity 14.
Although the effects of elevated lipid levels on MPS have not yet been fully elucidated, inflammation, oxidative stress, and neurological mechanisms are being implicated. High lipid levels may trigger inflammatory responses. Specifically, high- sensitivity C-reactive protein and phospholipase A2 levels have been reported to be higher in patients with MPS compared to healthy individuals 15. Lipid peroxidation is a marker of oxidative stress and may play a role in the pathophysiology of MPS. Recent studies have brought attention to the possibility that MPS may not be solely a local mechanical issue but may also be associated with systemic metabolic changes, particularly disorders in lipid metabolism 16. However, to the best of our knowledge, there is a significant lack of studies in the literature that directly investigate the relationship between dyslipidemia and MPS, as well as the impact of lipid levels on pain and other symptoms.
In this study, we aimed to investigate the prevalence of MPS in individuals with dyslipidemia and to examine the relationship between lipid levels and MPS symptoms.
Materials and Methods
This study included patients who presented to the Department of Internal Medicine and Physical Medicine and Rehabilitation of Adana City Training and Research Hospital and underwent lipid profile assessment. Medical records of 197 patients (132 females and 65 males) without inflammatory disease and with normal blood inflammatory markers [erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP)] were retrospectively reviewed.
Exclusion criteria were defined as the presence of inflammatory rheumatic diseases, inflammatory bowel diseases, a history of acute trauma or recent surgery, and myocardial infarction within the previous month. The diagnosis of myofascial pain syndrome (MPS) was established according to the diagnostic criteria described by Travell and Simons.
Data extracted from patient files included demographic characteristics (sex, age, weight, height, body mass index [BMI]), comorbidities, regular exercise habits, and Visual Analog Scale (VAS) scores. The severity of palpable muscle spasm was assessed using the four-point Pressure Pain Threshold Scale (PPTS), and only patients who underwent this evaluation were included in the study. Biochemical parameters, including serum triglycerides (TG), total cholesterol, high-density lipoprotein cholesterol (HDL-c), and low-density lipoprotein cholesterol (LDL-c) levels, were also recorded.
VAS and PPTS assessments, as well as lipid profile evaluations, were performed by the same physician to ensure consistency. All data were collected and verified through a comprehensive review of the patients’ medical records.
All participants were called to the laboratory after an average fasting period of 12 hours, and 5 mL of blood was drawn from the antecubital vein. The blood samples were transferred into plain tubes and allowed to clot. The samples were centrifuged at 4,000 rpm for 5 minutes at 4°C to separate the serum. Total cholesterol, TG, HDL-c, and LDL-c levels were measured in the separated serum. A Beckman Coulter AU5800 clinical chemistry autoanalyzer was used to measure the plasma lipid levels of individuals in the case and control groups. Measurements were performed using enzymatic colorimetric methods. The results were obtained in mg/dL.
Statistical MethodsStatistical analyses were performed using the SPSS v. 21 program. The demographic and clinical characteristics of the participants were summarized using means, standard deviations, medians, and minimum and maximum values. For group comparisons, the normality assumption was assessed using the Shapiro-Wilk and Kolmogorov-Smirnov tests. Differences between two independent groups were examined using the independent-sample t-test for normally distributed data and the Mann-Whitney U test for non-normally distributed data.
Logistic regression analysis was performed to predict dyslipidemia and MPS. In this analysis, the following predictors were used: dyslipidemia, MPS, age, sex, BMI, diabetes mellitus (DM), hypertension, and thyroid disease. The statistical adequacy of the model was determined by calculating the events per variable (EVP). This analysis included the events of the dyslipidemia group (175 cases). The independent variables in the logistic regression model were age, sex, BMI, DM, hypertension, thyroid disease, and MPS. The EVP was computed as follows:
EVP = total number of events (175) / number of independent variables (8) = 21.8
In the logistic regression analysis conducted to predict MPS, dyslipidemia, age, sex, BMI, DM, hypertension, and thyroid disease were identified as predictors (EPV = 19.3).
Ethical Approval This study was approved by the Ethics Committee of Adana City Training and Research Hospital (Date: 2024-10-10, No: 173).
Results
A total of 197 participants were evaluated for inclusion in this study. Among the 197 participants, 132 were female and 65 were male, with a mean age of 51.3 years (standard deviation: 11.48). Table 1 summarizes the demographic and clinical characteristics of the participants.
SD: standard deviation, BMI: body mass index, VAS: Visual Analog Scale, DM: diabetes mellitus, MPS: myofascial pain syndrome, CAD: coronary artery disease, PPTS: Pressure Pain Threshold Scale, LDL: low-density lipoprotein, HDL: high-density lipoprotein, TG: triglyceride.
The frequency of MPS and BMI was significantly higher in the dyslipidemia group than in the healthy control group (p < 0.01 for all) (Table 2).
When the participants were grouped according to their lipid levels (normal, borderline high, and high) and their clinical features were compared, the frequency of MPS was found to be higher in the borderline high group than in the healthy group. While VAS values were higher in the borderline high group than in the controls, PPTS scores differed across all three lipid groups (p < 0.05 for all) (Table 3).
In the logistic regression analysis conducted for dyslipidemia prediction, sex and MPS were found to be significant (p < 0.01 for both, odds ratio: 13.781 and 8.618, respectively). The model was statistically significant (χ2(8) = 31.546, p < 0.001), explaining 29.4% of the variance (R2CS = 0.148, R2N = 0.294,-2LL = 106.356).
Discussion
This study aimed to investigate the prevalence of MPS in dyslipidemia and the relationship between lipid levels and MPS symptoms. The results demonstrated that the frequency and severity of MPS increased in patients with dyslipidemia and that MPS could serve as a predictive factor for dyslipidemia. In this study, the prevalence of MPS in the dyslipidemic group was found to be significantly higher than in the group with normal lipid levels. This suggests a strong association between dyslipidemia and MPS and indicates a higher likelihood of developing MPS among individuals with dyslipidemia.
Lipids are biomolecules involved not only in energy metabolism but also in membrane stability, signal transduction, and inflammatory processes. It has been proposed that elevated serum lipid levels may trigger myofascial pain by inducing oxidative stress and inflammation in tissue 15.
In a study, the serum lipid profiles between patients with MPS and healthy individuals, no significant differences were found in total cholesterol, TG, HDL, LDL, or very low-density lipoprotein levels 7. However, another study published in the Abant Medical Journal reported that lipid levels in patients with MPS were higher than in healthy controls and that these levels were positively correlated with pain intensity and muscle spasm 16. Similarly, Ozgocmen and Ardicoglu showed that even borderline high lipid levels were associated with increased MPS and pain severity. They identified a significant relationship between total cholesterol and LDL levels and tissue compliance at trigger points, suggesting that MPS symptoms might undergo substantial changes even in the early stages of dyslipidemia 13. The findings of our study are consistent with the literature, indicating that the frequency and severity of MPS increase in dyslipidemia and that the presence of MPS may act as a predictor for dyslipidemia. These results support the hypothesis of a relationship between dyslipidemia and MPS and reinforce the notion that individuals with dyslipidemia are more likely to experience MPS. These findings suggest that dyslipidemia may influence musculoskeletal health and general pain experiences, which emphasizes the importance of considering pain in dyslipidemia management and underlines the need for a multidisciplinary approach.
This study also examined the relationship between dyslipidemia and MPS, as well as other demographic and clinical features, and showed that both the prevalence of MPS and BMI values were significantly higher in the dyslipidemia group compared to the healthy group, which is consistent with the literature 17. Nijs et al. provided evidence that chronic pain and inflammation might affect lipid metabolism, or conversely, that dyslipidemia might increase susceptibility to muscle pain through systemic inflammation and microvascular changes 18. Our findings not only suggest that dyslipidemia may contribute to the development of MPS but also reveal that the median PPTS score in the dyslipidemic group was higher than in the healthy group, indicating that pain and other MPS symptoms are more severe in individuals with dyslipidemia. These results underscore the importance of considering dyslipidemia in patients with MPS in clinical practice and highlight the relevance of lipid-lowering interventions in the management of pain.
In our study, the median BMI value in the dyslipidemic group was significantly higher than in the healthy group with normal lipid levels. This finding supports the strong association between dyslipidemia and overweight or obesity. The literature has emphasized that high BMI is a significant risk factor for dyslipidemia 19.
Although there is compelling evidence in the literature indicating a relationship between MPS and lipid levels, heterogeneity exists among study results. Variability may stem from differences in patient populations, sample sizes, and insufficient control of comorbid conditions affecting dyslipidemia (e.g., DM and obesity). Moreover, discrepancies in laboratory methods and diagnostic criteria used across studies contribute to this variability. Existing data suggest that abnormalities in lipid metabolism may play a role in the pathogenesis of MPS. However, it remains unclear whether this relationship is causal. Therefore, there is a need for prospective longitudinal studies and randomized controlled trials that assess the effects of lipid-lowering treatments on MPS symptoms.
Limitations
Among the strengths of our study is its comprehensive statistical examination of the relationship between MPS and dyslipidemia. Nonetheless, there are also limitations. As this is a cross-sectional study, causality cannot be established. Future prospective studies will be valuable in clarifying the direction of this relationship and elucidating underlying mechanisms. Moreover, including a larger sample size and incorporating diverse ethnic groups would enhance the generalizability of the findings. Lastly, stratification of dyslipidemia subtypes and the inclusion of inflammatory markers could provide deeper insight into the pathophysiology of this relationship.
Conclusion
This study demonstrated an association between dyslipidemia and both the prevalence and severity of MPS symptoms and further suggested that MPS could serve as a predictive factor for dyslipidemia. These results highlight the importance of considering MPS in the management of dyslipidemia and recognizing the presence of dyslipidemia in the evaluation and treatment of MPS and pain in clinical practice.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of Adana City Training and Research Hospital (Date: 2024-10-10, No: 173)
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
References
-
Lew J, Kim J, Nair P. Comparison of dry needling and trigger point manual therapy in patients with neck and upper back myofascial pain syndrome: a systematic review and meta-analysis. J Man Manip Ther. 2021;29(3):136-46.
-
Bennett RM. Myofascial pain syndromes and the fibromyalgia syndrome: a comparative analysis. In: Friction JR, Awad E, editors. Advances in pain research and therapy. 17th ed. New York: Raven Press; 1990.p.43-65.
-
Nadendla LK, Meduri V, Paramkusam G, Pachava KR. Evaluation of salivary cortisol and anxiety levels in myofascial pain dysfunction syndrome. Korean J Pain. 2014;27(1):30-4.
-
Kadi F, Waling K, Ahlgren C, et al. Pathological mechanisms implicated in localized female trapezius myalgia. Pain. 1998;78(3):191-6.
-
Sola AE, Bonica JJ. Myofascial pain syndromes. In: Bonica JJ, editor. The management of pain. 2nd ed. Pennsylvania: Lea and Febiger; 1990.p.352-67.
-
Shah JP, Danoff JV, Desai MJ, et al. Biochemicals associated with pain and inflammation are elevated in sites near to and remote from active myofascial trigger points. Arch Phys Med Rehabil. 2008;89(1):16-23.
-
Kaya A, Kamanlı A, Saitoğlu M, İlhan N. Miyofasiyal ağrı sendromlu hastaların serum lipit düzeyleri [Serum lipid levels in patients with myofascial pain syndrome]. Fırat Tıp Dergisi. 2006;11(3):160-2.
-
Hedayatnia M, Asadi Z, Zare-Feyzabadi R, et al. Dyslipidemia and cardiovascular disease risk among the MASHAD study population. Lipids Health Dis. 2020;16;19(1):42.
-
Obezite, Lipid Metabolizması ve Hipertansiyon çalışma grubu, editors. Dyslipidemia Diagnosis and Treatment Guidelines. Ankara: TEMD; 2021.p.17-8.
-
Klemp P, Halland AM, Majoos FL, Steyn K. Musculoskeletal manifestations in hyperlipidemia: a controlled study. Ann Rheum Dis. 1993;52(1):44-8.
-
Careless DJ, Cohen MG. Rheumatic manifestations of hyperlipidemia and antihyperlipidemia drug therapy. Semin Arthritis Rheum. 1993;23(2):90-8.
-
Struthers GR, Scott DL, Bacon PA, Walton KW. Musculoskeletal disorders in patients with hyperlipidemia. Ann Rheum Dis. 1983;42(5):519-23.
-
Ozgocmen S, Ardicoglu O. Lipid profile in patients with fibromyalgia and myofascial pain syndromes. Yonsei Med J. 2000;41(5):541-5.
-
Atzenı F, Alcıatı A, Salaffı F, et al.The association between body mass index and fibromyalgia severity: data from a cross-sectional survey of 2339 patients. Rheumatol Adv Pract. 2021;5(1):rkab015.
-
Shakouri SK, Dolatkhah N, Omidbakhsh S, Pishgahi A, Hashemian M. Serum inflammatory and oxidative stress biomarker levels are associated with pain intensity, pressure pain threshold and quality of life in myofascial pain syndrome. BMC Res Notes. 2020;13(1):510.
-
Tuna S, Alan S. Miyofasial ağrı sendromu ile kan lipid düzeyleri arasındaki ilişki [Relationship between myofascial pain syndrome and blood lipid levels]. Abant Medical Journal. 2015;4(1):44-6.
-
Wijma AJ, van Wilgen CP, Meeus M, Nijs J. Clinical biopsychosocial physiotherapy assessment of patients with chronic pain: The first step in pain neuroscience education. Physiother Theory Pract. 2016;32(5):368-84.
-
Nijs J, Van Houdenhove B, Van Oosterwijck J. Recognition of central sensitization in patients with musculoskeletal pain: application of pain neurophysiology in manual therapy practice. Manual Therapy. 2010;15(2):135-41.
-
Ferrières J, Lautsch D, Gitt AK, et al. Body mass index impacts the choice of lipid-lowering treatment with no correlation to blood cholesterol - Findings from 52 916 patients in the Dyslipidemia International Study (DYSIS). Diabetes Obes Metab. 2018;20(11):2670-4.
Tables
Table 1. Demographic and clinical characteristics of the participants
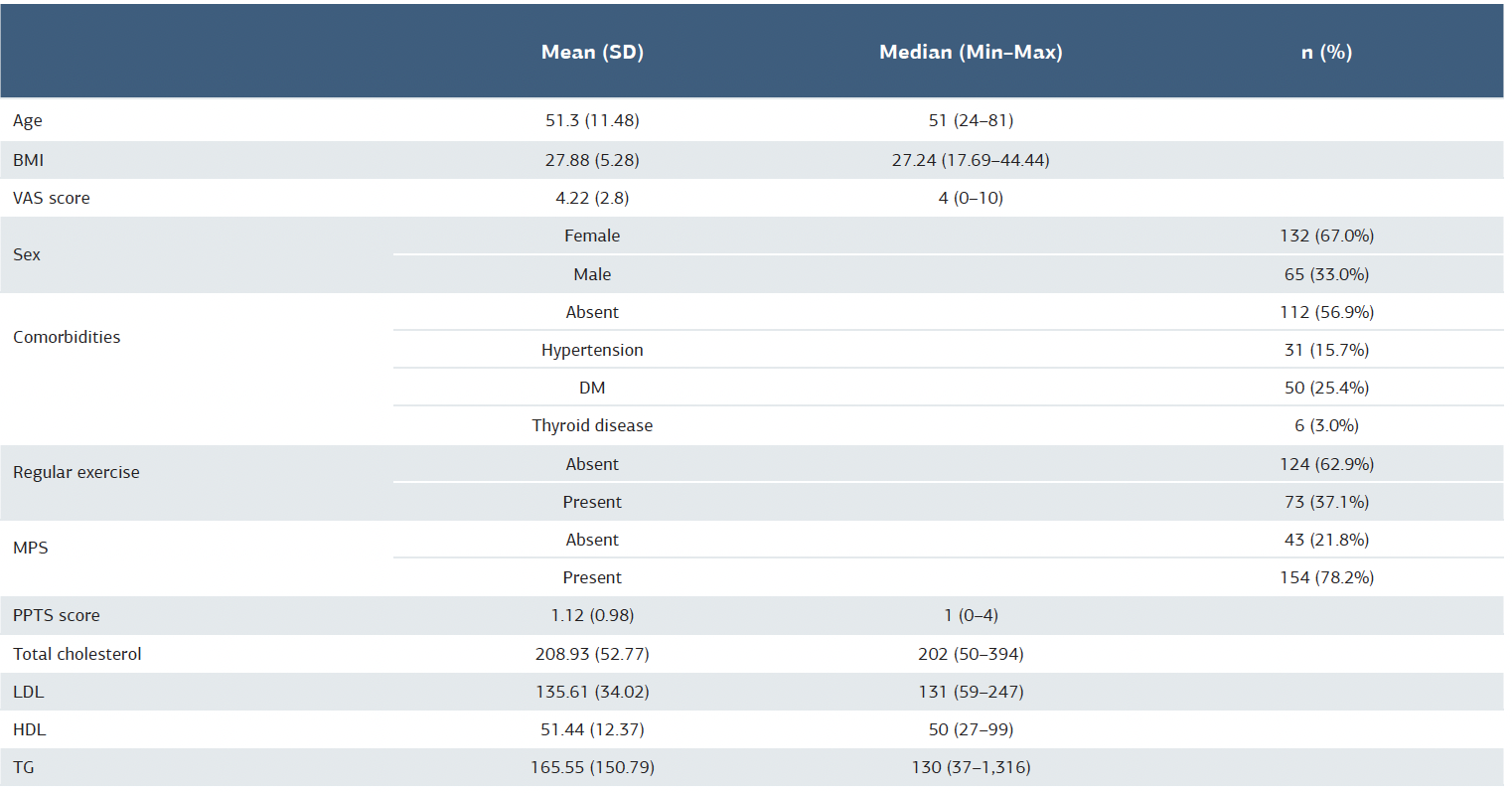
SD = standard deviation; BMI = body mass index; VAS = Visual Analog Scale; DM = diabetes mellitus; MPS = myofascial pain syndrome; CAD = coronary artery disease; PPTS = Pressure Pain Threshold Scale; LDL = low-density lipoprotein; HDL = high-density lipoprotein; TG = triglyceride
Table 2. Comparison of demographic and clinical characteristics between dyslipidemia and healthy control groups
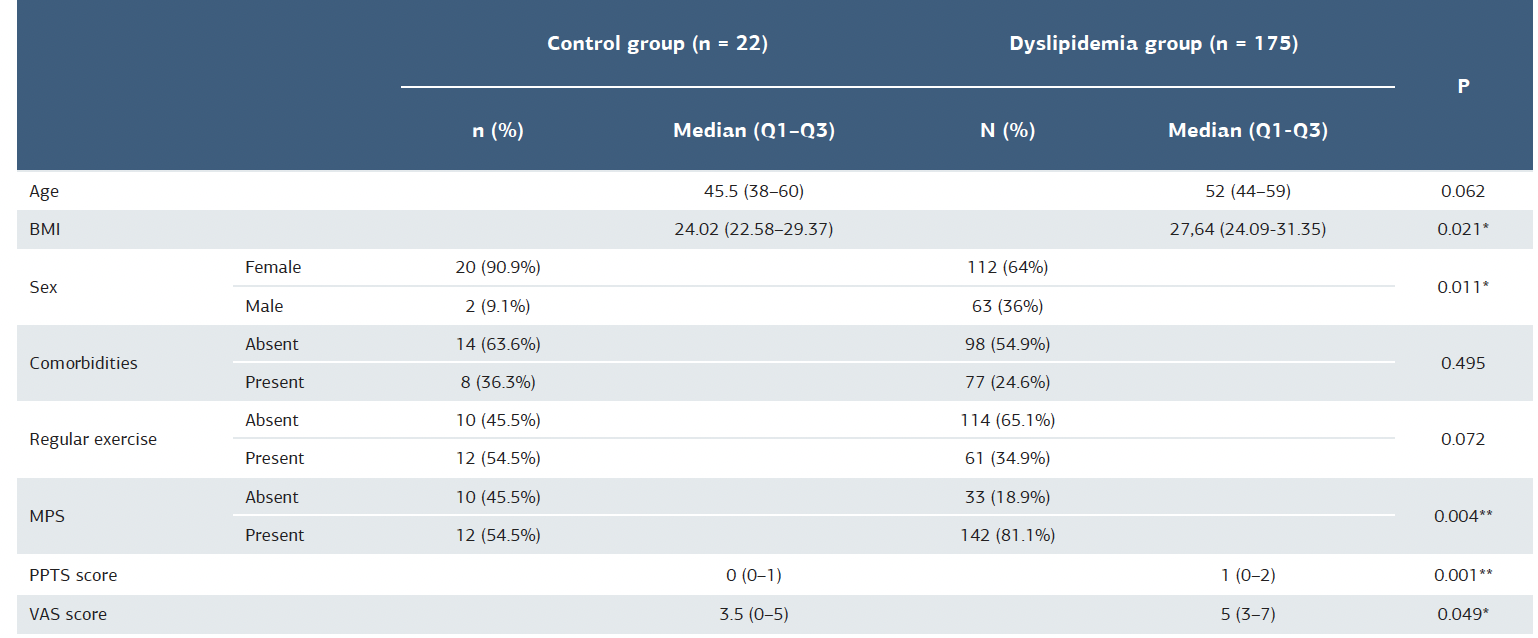
BMI = body mass index; MPS = myofascial pain syndrome; PPTS = Pressure Pain Threshold Scale; VAS = Visual Analog Scale * = p<0.05; ** = p<0.01
Table 3. Comparison of clinical features according to lipid levels
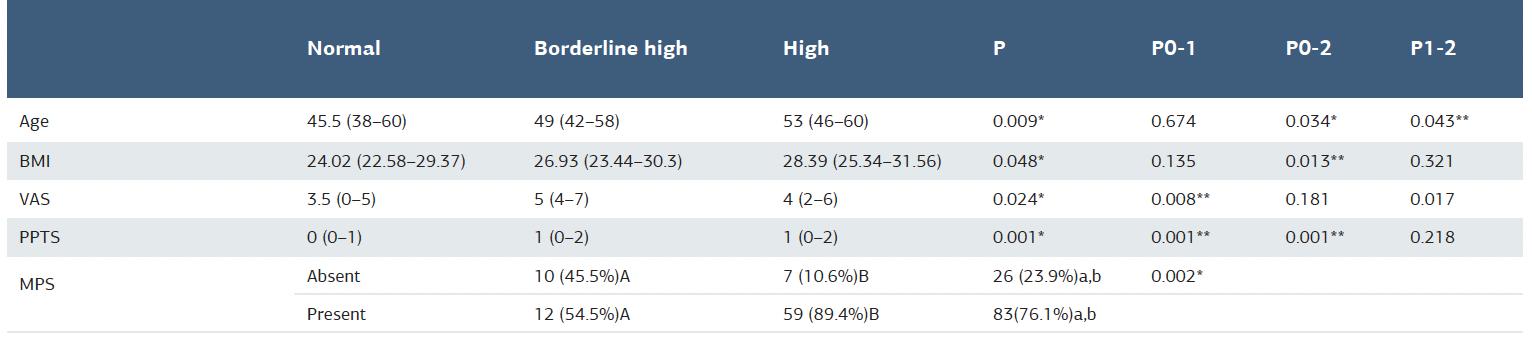
*Statistically significant at p < 0.05, **Statistically significant at p < 0.017 (Bonferroni correction) BMI: body mass index, PPTS = Pressure Pain Threshold Scale; VAS = Visual Analog Scale; MPS = myofascial pain syndrome
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Nilüfer Aygün Bilecik, Serpil Demir, Yunus Çoşkun, Sıdıka Büyükvural Şen, Meryem Kösehasanoğulları. Can Dyslipidemia be a significant factor in myofascial pain syndrome? Ann Clin Anal Med 2025;16(10):747-751

