Artemisinin suppresses proliferation and induces oxidative damage and apoptosis in H1395 lung adenocarcinoma cells
Artemisinin effects on H1395 lung adenocarcinoma
Authors
Abstract
Aim Lung cancer is the most lethal type of cancer worldwide. Particularly in the metastatic stage, the cure and overall survival rates of non-small cell lung cancer (NSCLC) remain low. Considering the limitations of current therapeutic options, alternative and naturally derived compounds such as artemisinin (ART) are worth investigating for their potential role in lung cancer treatment. In the present study, we evaluated the effect of artemisinin on cell proliferation, oxidative stress, and apoptosis in lung adenocarcinoma cells.
Methods In this study, the antiproliferative effect of ART on H1395 lung cancer cells was assessed using an MTT assay. Oxidative damage was evaluated using 8-Hydroxy-2’-deoxyguanosine (8-OHdG) levels, total antioxidant status (TAS), and total oxidant status (TOS), while apoptosis was analyzed using the caspase-3 ELISA method. Additionally, total RNA was isolated from both the control and treated groups, followed by cDNA synthesis, and mRNA expression levels of apoptosis-related genes were examined using real time-PCR (RT-PCR).
Results The viability of H1395 lung cancer cells decreased in time-and dose- dependent manners following ART treatment. The IC₅₀ value of ART at 72 h was 1167 µM. ART treatment reduced TOS and increased TAS, resulting in a decreased oxidative stress index (OSI) in H1395 cells. However, the changes in 8-OHdG and caspase-3 levels, as measured by ELISA, were not statistically significant. Similarly, no significant differences were observed in the mRNA expression levels of Bax, Bcl-2, caspase-3, caspase-7, caspase-8, and caspase-9 between the control and ART-treated groups.
Conclusion ART shows potential as an anticancer agent and contributes to the development of novel therapeutic approaches for lung cancer.
Keywords
Introduction
Lung cancer was identified as the most frequently diagnosed cancer type in 2022 and remains the leading cause of cancer-related mortality to date. Over the past two decades, numerous novel therapeutic agents have been introduced for its treatment.1 Our knowledge of lung cancer biology and the processes that drive tumor progression has greatly improved. The application of innovative therapeutic approaches, such as tyrosine kinase inhibitors and immunotherapies, has demonstrated notable survival benefits in selected patient groups. Nevertheless, cure rates and overall survival rates for non-small cell lung cancer (NSCLC), particularly in the metastatic stage, remain low.2
Despite progress in chemotherapy, targeted therapies, and immunotherapy, treatment-related toxicity and adverse effects continue to represent major limitations in lung cancer management. Consequently, there is a growing need for the development of novel bioactive compounds that not only exert effective anticancer activity but also minimize toxicity.3,4
Considering the limitations of current therapeutic options, the investigation of alternative and naturally derived compounds such as artemisinin (ART) holds scientific significance in the search for new treatment strategies against lung cancer. ART, a compound traditionally used in the treatment of malaria, has recently gained attention owing to its anticancer properties.5,6 Preclinical studies have demonstrated that ART and its derivatives inhibit cell proliferation, induce apoptosis, and selectively target tumor cells, particularly in various solid tumors, through mechanisms involving oxidative stress.7 In vitro studies conducted on NSCLC cell lines have shown that the cytotoxic effects of ART increase in a dose- and time-dependent manner.8
The mechanistic basis underlying the effects of ART on lung cancer cells has not been fully delineated, and its exact biological effects remain unclear. This study aimed to comprehensively investigate the antiproliferative, anticancer, and apoptosis-inducing effects of Artemisinin on the H1395 non-small cell lung cancer cell line under in vitro conditions. As one of the first comprehensive studies conducted specifically on this cell line, this study is expected to provide valuable insights that may guide the development of future therapeutic strategies.
Materials and Methods
Cell CultureART was purchased from Sigma-Aldrich (USA). H1395 lung cancer cells (The H1395 cell line was obtained from Dr. Şakir Akgün, Kafkas University, Kars, Türkiye) were cultured in RPMI-1640 medium (Gibco Laboratories, Grand Island, NY) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Capricorn Scientific GmbH), 20 μg/ml streptomycin, 20 U/ml penicillin, 1 mM sodium pyruvate (Biological Industries, Israel), and 0.1 mM amino acid solution (Biological Industries, Israel). Cells were cultured at 37 °C in a humidified incubator under an atmosphere of 5% CO₂. To assess the antiproliferative effects of ART, cells were exposed to increasing concentrations (20, 40, 80, 160, 320, 640, 1000, 1280, and 1500 µM) of ART for 24, 48, and 72 h in a time- and dose-dependent manner.
MTT Cell Proliferation AssayThe antiproliferative activity of ART on H1395 lung cancer cells was evaluated using the MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium) assay. Cells were seeded at a density of 5 × 10³ cells per well in 96-well plates, and the assay was performed according to the manufacturer’s instructions (Cell Proliferation Kit; GoldBio, USA). Following completion of the treatment intervals, MTT reagent was added as recommended by the manufacturer, and the absorbance was measured spectrophotometrically. The optical density at 570 nm was recorded using a Biotek Epoch 2 microplate reader with the Gen5 software. The IC₅₀ concentration determined in these assays was used as the treatment dose for subsequent molecular experiments.
Cell Viability (%) = ([Treatment group OD570 - Blank well OD570 ]/[Untreated group OD570 - Blank well OD570 ]) × 100%.
Total Antioxidant Status, Total Oxidant Status, and Oxidative Stress Index Assay The effects of ART on TAS and TOS in H1395 lung cancer cells were assessed using Rel Assay TAS and TOS kits (Rel Assay Kit Diagnostics, Gaziantep, Türkiye), following the manufacturer’s instructions. TAS and TOS measurements were performed using a Biotek Epoch 2 microplate reader and Gen5 software, respectively. The TAS and TOS values were calculated using the corresponding formulas provided by the kit.
TOS= ([ΔabsSample]/[ΔabsStandard] X Concentration of standard
TAS= ([ΔAbs H2O] - [ΔAbs Sample]/[ΔAbs H2O] - [ΔAbs Standard])
OSI serves as a measure of oxidative stress levels and is a unitless parameter calculated by taking the ratio of TOS to TAS.9 To facilitate this calculation, TAS was initially converted from mmol Trolox equivalents per liter to µmol Trolox equivalents per liter.
OSI = ([TOS, µmol H2O2 equivalents/L]/[TAS, µmol Trolox equivalents/L] × 100)
8-hydroxy-2’-Deoxyguanosine AssayThe human 8-OHdG biomarker, which is indicative of DNA damage resulting from oxidative stress, was measured using the ELISA method.10 The assay was performed using an 8-OHdG kit (Bioassay Technology Laboratory, BT Lab, Shanghai, China) according to the manufacturer’s instructions and protocols.
Caspase-3 AssayThe protein level of caspase-3 in ART-treated H1395 lung cancer cells was measured using an ELISA kit from the Bioassay Technology Laboratory (BT Lab, Shanghai, China) following the manufacturer’s protocols.
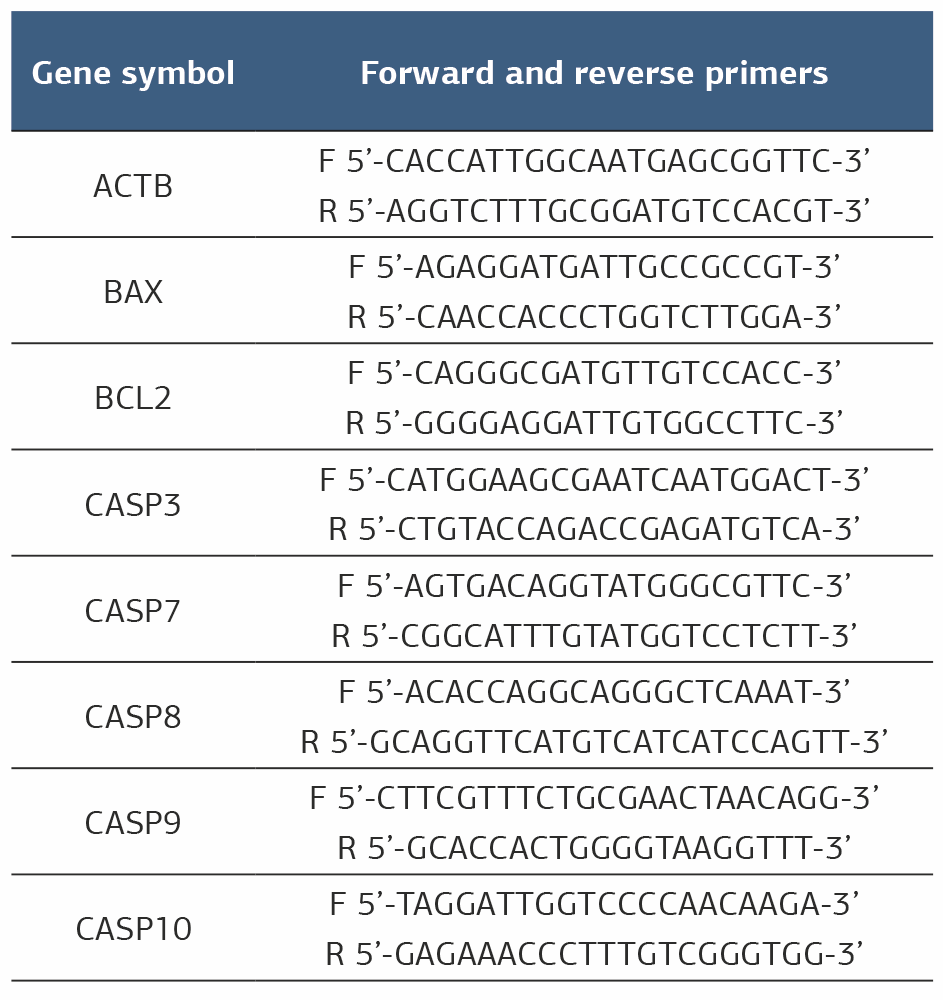
Total RNA Isolation, cDNA Synthesis, and RT-PCR AnalysisRNA extraction was conducted using Trizol reagent (Invitrogen, USA) in accordance with the supplier’s recommendations, and cDNA generation was performed using the A.B.T. kit with RNase inhibitor (A.B.T., Türkiye). Changes in the mRNA expression levels of Bax (BCL2-associated X), Bcl-2 (BCL-2 apoptosis regulator), caspase-3 (casp-3), caspase-7 (casp-7), caspase-8 (casp-8), and caspase-9 (casp-9) were evaluated by Real-Time PCR (RT-PCR) using the Rotor-Gene Q system (Qiagen, Hilden, Germany). Βeta-Actin (ACTB) was used for normalization. Primer sequences used in this study are listed in Table 1. Real-time PCR was conducted following the SYBR Green qPCR Master Mix protocol using ABTTM 2X qPCR SYBR-Green Master Mix (Türkiye).
Ethical ApprovalThis study was conducted using a commercially available human lung adenocarcinoma cell line (H1395). According to the decision of the Non-Interventional Research Ethics Committee of Ordu University (Date: 2025-07-18, No: 253), ethical approval was not required for this in vitro experimental study.
Statistical AnalysisAll statistical analyses were performed using GraphPad Prism version 9.4.1 (GraphPad Software, San Diego, CA, USA). RT-PCR quantification was performed using the 2^−ΔΔCT method, with further analysis using the GeneGlobe RT-PCR Analysis RT2 Profile PCR Array Data Analysis platform (Qiagen). Values are reported as mean ± S.D. Statistical differences among multiple groups were evaluated using analysis of variance (ANOVA), followed by Dunnett’s post-hoc test for comparisons of each group’s mean with the control. For pairwise analyses, either an unpaired t-test or Mann–Whitney U test was applied, depending on the data distribution. A p-value of < 0.05 was considered statistically significant (ns p > 0.05, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001).
Reporting GuidelinesThis in vitro experimental study was conducted in accordance with standard laboratory research principles. No specific reporting guideline was applicable.
Results
MTT AnalysisUpon ART treatment, the viability of H1395 cells was assessed using the MTT assay. A decrease in cell viability was observed in time- and dose-dependent manners. The half maximal inhibitory concentration (IC50) dose of ART was determined to be 1167 µM at the 72nd hour in the H1395 cell line (Figure 1).
TAS, TOS, and OSI AnalysisTo evaluate the effect of ART on oxidative stress in H1395 lung cancer cells, TAS, TOS, and OSI were measured. ART treatment led to a decrease in TAS and increase in TOS levels (TAS: Control, 1.237 ± 0.059; ART-treated, 1.176 ± 0.056; p = 0.66). TOS: Control, 5.949 ± 0.895; ART-treated, 6.203 ± 0.895; p = 0.66). The OSI increased in the ART-treated group compared to the control in H1395 lung cancer cells (control: 0.483 ± 0.095; ART-treated: 0.530 ± 0.101; p = 0.66) (Figure 2). However, these differences were not statistically significant.
8-OHdG Analysis
8-OHdG levels were measured using ELISA. Changes in protein concentration were determined by comparing ART-treated H1395 lung cancer cells with the control group. According to the data, the mean 8-OHdG concentration in the H1395 control group was 0.805 ± 0.064 ng/mL, while in the cells treated with ART, it was 0.809 ± 0.158 ng/mL. When the results were compared, no significant differences were found (p > 0.99, Figure 3).
Caspase-3 AnalysisThe possible caspase-3 activation by ART in H1395 lung cancer cells was analyzed by ELISA. Accordingly, the mean caspase-3 levels in the serum were 0.791 ± 0.022 for the control group and 0.793 ± 0.054 for ART-treated H1395 cells. When the results were compared statistically, no significant differences were found (p > 0.99, Figure 3).
RT-PCR AnalysisChanges in the mRNA expression of apoptosis-related genes were detected by real-time PCR. In the group treated with the IC50 dose of ART, no statistically significant changes were observed in the expression levels of Bax (0.36, p = 0.178), caspase-3 (0.95, p = 0.392), caspase-7 (0.56, p = 0.387), caspase-8 (15.71, p = 0.372), caspase-9 (0.11, p = 0.368), or caspase-10 (0.12, p = 0.111) compared to the control group. However, a statistically significant change in Bcl-2 expression (0.13, p = 0.042*) was detected.
Discussion
Lung cancer is the leading cause of cancer-related deaths globally. In this study, we showed that artemisinin exerts pronounced in vitro effects on lung cancer cells, highlighting its potential as a basis for future therapeutic interventions. Artemisinin is known to inhibit pathological processes that promote cancer progression in the tumor microenvironment owing to its anti-inflammatory and antioxidant properties. Its low toxicity and minimal effects on healthy cells make it a promising candidate for chemotherapy-resistant lung cancer subtypes. Taken together, these properties suggest that artemisinin is a potentially valuable phytochemical for experimental and preclinical studies of lung cancer. Importantly, our study is among the first to investigate the effects of artemisinin on lung cancer cells, addressing a notable gap in the current literature and providing a foundation for future research on novel therapeutic strategies.
Li et al. investigated the effects of ART and its derivatives on the viability of A549 lung cancer cells using an MTT assay. In their study, ART was administered at concentrations of 50, 100, 200, 250, 500, 750, and 1000 µM, and the IC₅₀ value was determined to be 769.60 µM.15 In a study by Hu et al., the artemisinin derivative, artesunate, was investigated for its antiproliferative effects on A549 and H1299 lung cancer cell lines, and the IC₅₀ values were reported as 28.8 µg/mL and 27.2 µg/mL, respectively.11
Lu et al. determined the IC₅₀ value of artemisinin in the HCC827 lung cancer cell line to be 56.1 µM, demonstrating its inhibitory effect on cell viability and proliferation.12 Conjugated forms of artemisinin and its derivatives have also been used in in vitro studies to enhance their bioavailability. An example of this is the study conducted by Kumar et al. In this study, the conjugated form of dihydroartemisinin with hyaluronic acid was applied to the A549 lung cancer cell line, demonstrating antiproliferative effects at lower doses.13 In our study, ART significantly reduced the viability of H1395 lung cancer cells in a time- and dose-dependent manner, with an IC₅₀ of 1167 µM at 72 h, supporting its potential as an anticancer agent.
Oxidative stress is recognized as a key factor in cancer development, as it can disrupt DNA integrity, alter gene regulation, and interfere with cell-to-cell communication. These molecular alterations may drive abnormal cell proliferation and contribute to genomic instability, thereby playing a central role in the initiation and progression of malignancies. Oxidative stress may also trigger cell death pathways, such as apoptosis and ferroptosis, underscoring its dual function in the context of cancer biology.14,15
Ning et al. demonstrated in vivo that dihydroartemisinin alleviates radiation-induced pathological cell damage and histologically reduces TNF-α and TGF-β levels following irradiation.16
In a study by Ho and colleagues, the effects of artesunate on allergic asthma were investigated experimentally.17 They demonstrated a significant reduction in oxidative damage markers, such as 8-OHdG and 3-nitrotyrosine. Li et al. demonstrated the effects of ART and its derivative artesunate on DNA damage in the A549 lung cancer cell line using the comet assay.8 In our study, ART treatment caused minor changes in TAS, TOS, OSI, and 8-OHdG levels in H1395 lung cancer cells; however, these differences were not statistically significant.
Apoptosis is a tightly regulated form of programmed cell death that plays a vital role in maintaining tissue homeostasis and eliminating damaged, mutated, or potentially malignant cells. In healthy tissues, it serves as a defense mechanism to prevent uncontrolled cell proliferation and tumor formation. Dysregulation of apoptotic pathways is a hallmark of cancer and contributes to tumor survival, resistance to therapy, and disease progression. Therefore, many anticancer therapies aim to restore or enhance apoptotic signaling in malignant cells.18,19
Caspases are important proteolytic enzymes of the cysteine protease family, which play critical roles in apoptosis and inflammation. If caspase activity is disrupted, various health problems such as neurodegenerative diseases, autoimmune disorders, and cancer may arise. A decrease in or inhibition of caspase activity in cancer cells contributes to resistance to cell death and promotes tumor progression. Therefore, reactivation of caspases or stimulation of caspase pathways can enhance the treatment efficacy by inducing apoptosis and eliminating cancer cells. Additionally, in some novel therapeutic approaches, caspases play a role in inducing pyroptosis, an inflammatory form of cell death, which helps strengthen the immune response within the tumor microenvironment. For these reasons, caspases are of great importance in cancer therapy both as target molecules and as biomarkers that enhance treatment response.20,21 In their study, Ganguli et al. showed that treatment of A549 cells with ART induced cytochrome C release, leading to caspase-3-dependent cell death. The same study also demonstrated that scavenging reactive oxygen species with antioxidants inhibited caspase-3 activity and rescued the cells from apoptosis.22 Lu et al. demonstrated that dihydroartemisinin, a derivative of artemisinin, induces apoptosis by activating caspase-3 in ASTC-a-1 lung cancer cells, as shown by the annexin V assay.23 Using flow cytometry, Lu et al. demonstrated that ART increased the apoptosis rate in HCC827 lung cancer cells.12 The same study also reported that ART treatment upregulated caspase-3 gene expression in this cell line. Our study demonstrated that caspase-3 levels did not significantly differ between ART-treated and control H1395 cells. This suggests that ART can induce apoptosis through different pathways under the tested conditions, but it also suggests that ART derivatives may exert their effects through different death pathways.
In our study, changes in the expression of apoptosis-related genes, including Bax, Bcl-2, caspase-3, caspase-7, caspase-8, caspase-9, and caspase-10, were evaluated. Although a 15.71-fold increase was observed in caspase-8 levels, the difference was not statistically significant. In contrast, upregulation of the Bcl-2 gene expression was statistically significant. Bcl-2 is an anti-apoptotic protein that regulates the permeability of the mitochondrial outer membrane and prevents cytochrome c release, thereby inhibiting the intrinsic (mitochondrial) apoptotic pathway and preventing cell death. Its overexpression in cancer cells is closely associated with therapeutic resistance. Caspase-8, a key initiator of the extrinsic apoptotic pathway, is activated upon stimulation of death receptors such as Fas and TRAIL-R. Activated caspase-8 directly triggers effector caspases, including caspase-3, to induce apoptosis and engages the intrinsic pathway via Bid, facilitating the synergistic progression of cell death.24,25
Limitations
This study has some limitations. The findings were analyzed in a lung adenocarcinoma cell line (H1395). This situation may limit the generalizability to tumor models, as it prevents the potential variations among other NSCLC cell subtypes from being fully clarified. However, since all analyses were performed under in vitro conditions, the observed effects of ART may not fully reflect its in vivo biological activity, toxicity, and bioavailability. For all these reasons, future studies should include dose optimization in different lung cell lines, comparisons with ART and its derivatives, and experimental validation approaches to strengthen the biological significance and reliability of the findings.
Conclusion
In conclusion, our findings demonstrate that ART reduces the viability of H1395 lung adenocarcinoma cells in a time- and dose-dependent manner. Although ART treatment was associated with changes in oxidative stress parameters and apoptosis-related gene expression, most of these alterations were not statistically significant under the tested conditions. These results suggest that ART may exert antiproliferative effects in this cell line; however, further mechanistic and in vivo studies are required to clarify its potential role as an anticancer agent.
Declarations
Ethics Declarations
This study was conducted using a commercially available human lung adenocarcinoma cell line (H1395). It did not involve human participants or animal subjects; therefore, ethical approval and informed consent were not required.
Animal and Human Rights Statement
Not applicable. This study was conducted using a commercially available human cancer cell line and did not involve human participants or animal subjects.
Informed Consent
Not applicable. This study did not involve human participants or identifiable personal data.
Data Availability
Data are contained within the article.
Conflict of Interest
The authors declare that there is no conflict of interest
Funding
None.
Author Contributions (CRediT Taxonomy)
Conceptualization: S.K., G.G.
Methodology: G.G., M.S.
Formal Analysis: M.S.
Investigation: S.K., G.G.
Data Curation: M.S.
Writing – Original Draft Preparation: S.K.
Writing – Review & Editing: G.G., M.S.
Supervision: G.G.
Scientific Responsibility Statement
The authors declare that they are responsible for the scientific content of this manuscript, including the study design, data collection, analysis and interpretation of data, drafting and revising the manuscript, and approval of the final version.
Abbreviations
ACTB: beta-actin
ANOVA: analysis of variance
ART: artemisinin
cDNA: complementary DNA
CO₂: carbon dioxide
CT: cycle threshold
ELISA: enzyme-linked immunosorbent assay
FBS: fetal bovine serum
IC₅₀: half maximal inhibitory concentration
mRNA: messenger RNA
NSCLC: non-small cell lung cancer
OD: optical density
OSI: oxidative stress index
PCR: polymerase chain reaction
RT-PCR: real-time polymerase chain reaction
SD: standard deviation
TAS: total antioxidant status
TGF-β: transforming growth factor beta
TNF-α: tumor necrosis factor alpha
TOS: total oxidant status
References
-
Jeon H, Wang S, Song J, Gill H, Cheng H. Update 2025: management of non small-cell lung cancer. Lung. 2025;203(1):53. doi:10.1007/S00408-025-00801-X
-
Bilgin B, Sendur MAN, Yucel S, et al. Real-life comparison of the afatinib and first-generation tyrosine kinase inhibitors in nonsmall cell lung cancer harboring EGFR exon 19 deletion: a Turk Oncology Group (TOG) study. J Cancer Res Clin Oncol. 2021;147(7):2145-2152. doi:10.1007/S00432-020-03501-6
-
Aybek SD, Secme M, Ilhan H, Acik L, Celik SP, Gulbay G. Multifaceted applications of zerumbone-loaded metal-organic framework-5: anticancer, antibacterial, antifungal, DNA-binding, and free radical scavenging potentials. Molecules. 2025;30(14):2936. doi:10.3390/MOLECULES30142936
-
Wei Z, Chen J, Zuo F, et al. Traditional Chinese medicine has great potential as candidate drugs for lung cancer: a review. J Ethnopharmacol. 2023;300:115748. doi:10.1016/j.jep.2022.115748
-
Feng X, Cao S, Qiu F, Zhang B. Traditional application and modern pharmacological research of Artemisia annua L. Pharmacol Ther. 2020;216:107650. doi:10.1016/j.pharmthera.2020.107650
-
Mölleken J, Kragl A, Monecke A, Metelmann I, Krämer S, Kallendrusch S. Artemisinin derivatives differently affect cell death of lung cancer subtypes by regulating GPX4 in patient-derived tissue cultures. Cell Death Discov. 2025;11(1):256. doi:10.1038/S41420-025-02537-2
-
Zhang Q, Yi H, Yao H, et al. Artemisinin derivatives ınhibit non-small cell lung cancer cells through ınduction of ROS-dependent apoptosis/ferroptosis. J Cancer. 2021;12(13):4075-4085. doi:10.7150/JCA.57054
-
Li X, Gu S, Sun D, Dai H, Chen H, Zhang Z. The selectivity of artemisinin-based drugs on human lung normal and cancer cells. Environ Toxicol Pharmacol. 2018;57:86-94. doi:10.1016/j.etap.2017.12.004
-
Acar A, Ugur Cevik M, Evliyaoglu O, et al. Evaluation of serum oxidant/antioxidant balance in multiple sclerosis. Acta Neurol Belg. 2012;112(3):275-280. doi:10.1007/S13760-012-0059-4
-
Seçme M, Aybek SD, Gülbay G, Dodurga Y. The effect of thymoquinone on cell proliferation, 8-hydroxy-2 -deoxyguanosine level and expression changes of DNA repair and oxidative stress-related genes in MCF-7 breast cancer cells. Pamukkale Tıp Dergisi. 2025;18(2):304-312. doi:10.31362/PATD.1592341
-
Hu P, Ni C, Teng P. Effects of artesunate on the malignant biological behaviors of non-small cell lung cancer in human and its mechanism. Bioengineered. 2022;13(3):6590-6599. doi:10.1080/21655979.2022.2042141
-
Lu Z, Jiang J, Yao X, Hou G. Network pharmacological mechanism and molecular experimental validation of artemisinin in the treatment of lung adenocarcinoma. Toxicol Appl Pharmacol. 2025;495:117226. doi:10.1016/j.taap.2025.117226
-
Kumar R, Singh M, Meena J, et al. Hyaluronic acid - dihydroartemisinin conjugate: synthesis, characterization and in vitro evaluation in lung cancer cells. Int J Biol Macromol. 2019;133:495-502. doi:10.1016/j.ijbiomac.2019.04.124
-
Li K, Deng Z, Lei C, Ding X, Li J, Wang C. The role of oxidative stress in tumorigenesis and progression. Cells. 2024;13(5):441. doi:10.3390/CELLS13050441
-
Jelic MD, Mandic AD, Maricic SM, Srdjenovic BU. Oxidative stress and its role in cancer. J Cancer Res Ther. 2021;17(1):22-28. doi:10.4103/JCRT.JCRT_862_16
-
Ning X, Zhao W, Wu Q, Wang C, Liang S. Therapeutic potential of dihydroartemisinin in mitigating radiation-induced lung injury: Inhibition of ferroptosis through Nrf2/HO-1 pathways in mice. Immun Inflamm Dis. 2024;12(2):e1175. doi:10.1002/IID3.1175
-
Ho WE, Cheng C, Peh HY, et al. Anti-malarial drug artesunate ameliorates oxidative lung damage in experimental allergic asthma. Free Radic Biol Med. 2012;53(3):498-507. doi:10.1016/j.freeradbiomed.2012.05.021
-
Morana O, Wood W, Gregory CD. The apoptosis paradox in cancer. Int J Mol Sci. 2022;23(3):1328. doi:10.3390/IJMS23031328
-
Moyer A, Tanaka K, Cheng EH. Apoptosis in cancer biology and therapy. Annu Rev Pathol. 2025;20(1):303-328. doi:10.1146/annurev-pathmechdis-051222-115023
-
Allani M, Akhilesh, Tiwari V. Caspase-driven cancer therapies: navigating the bridge between lab discoveries and clinical applications. Cell Biochem Funct. 2024;42(2):e3944. doi:10.1002/CBF.3944
-
Gao M, Sun Q, Zhang H, et al. Bioinspired nano-photosensitizer-activated caspase-3/GSDME pathway induces pyroptosis in lung cancer cells. Adv Healthc Mater. 2024;13(26):e2401616. doi:10.1002/adhm.202401616
-
Ganguli A, Choudhury D, Datta S, Bhattacharya S, Chakrabarti G. Inhibition of autophagy by chloroquine potentiates synergistically anti-cancer property of artemisinin by promoting ROS dependent apoptosis. Biochimie. 2014;107(Pt B):338-349. doi:10.1016/j.biochi.2014.10.001
-
Lu YY, Chen TS, Qu J Le, Pan WL, Sun L, Wei X Bin. Dihydroartemisinin (DHA) induces caspase-3-dependent apoptosis in human lung adenocarcinoma ASTC-a-1 cells. J Biomed Sci. 2009;16(1):16. doi:10.1186/1423-0127-16-16
-
Abdulkareem SJ, Jafari-Gharabaghlou D, Farhoudi-Sefidan-Jadid M, Salmani-Javan E, Toroghi F, Zarghami N. Co-delivery of artemisinin and metformin via PEGylated niosomal nanoparticles: potential anti-cancer effect in treatment of lung cancer cells. Daru. 2024;32(1):133-144. doi:10.1007/S40199-023-00495-7
-
Zhao Y, Liu J, Liu L. Artesunate inhibits lung cancer cells via regulation of mitochondrial membrane potential and induction of apoptosis. Mol Med Rep. 2020;22(4):3017-3022. doi:10.3892/MMR.2020.11341
Figures
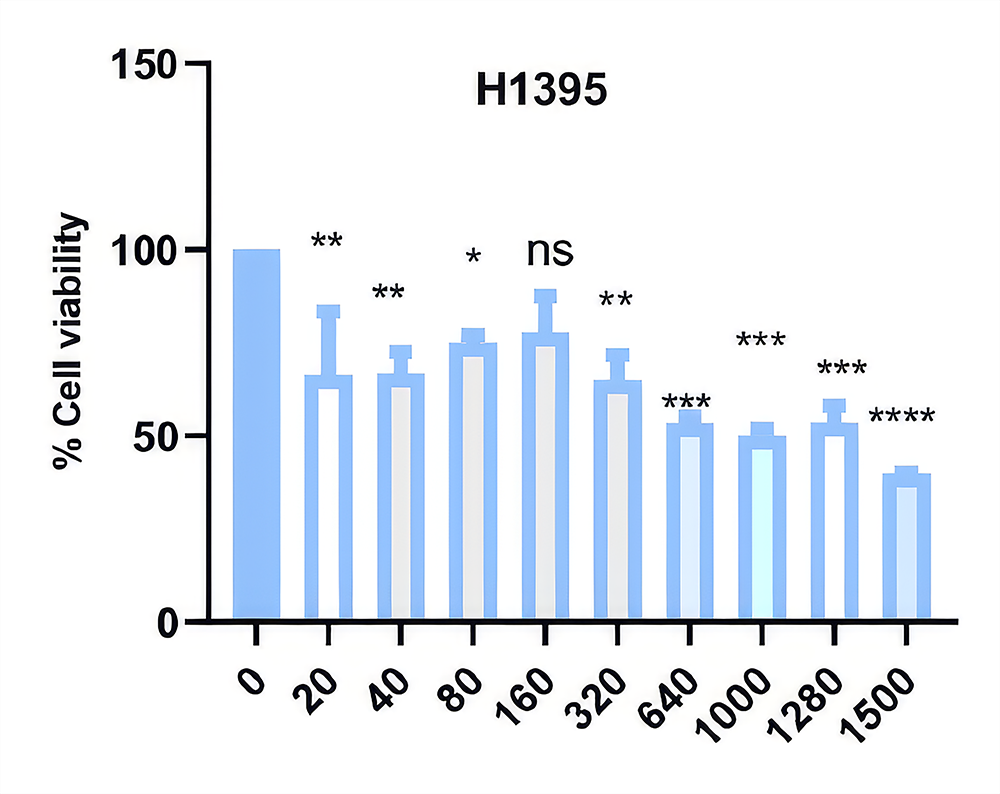
Figure 1. H1395 lung cancer cells were treated with ART at different concentrations and time intervals, and their viability was assessed by MTT assay. IC50 doses of ART in H1395 lung cancer cells were detected as 1167 µM at the 72nd hour
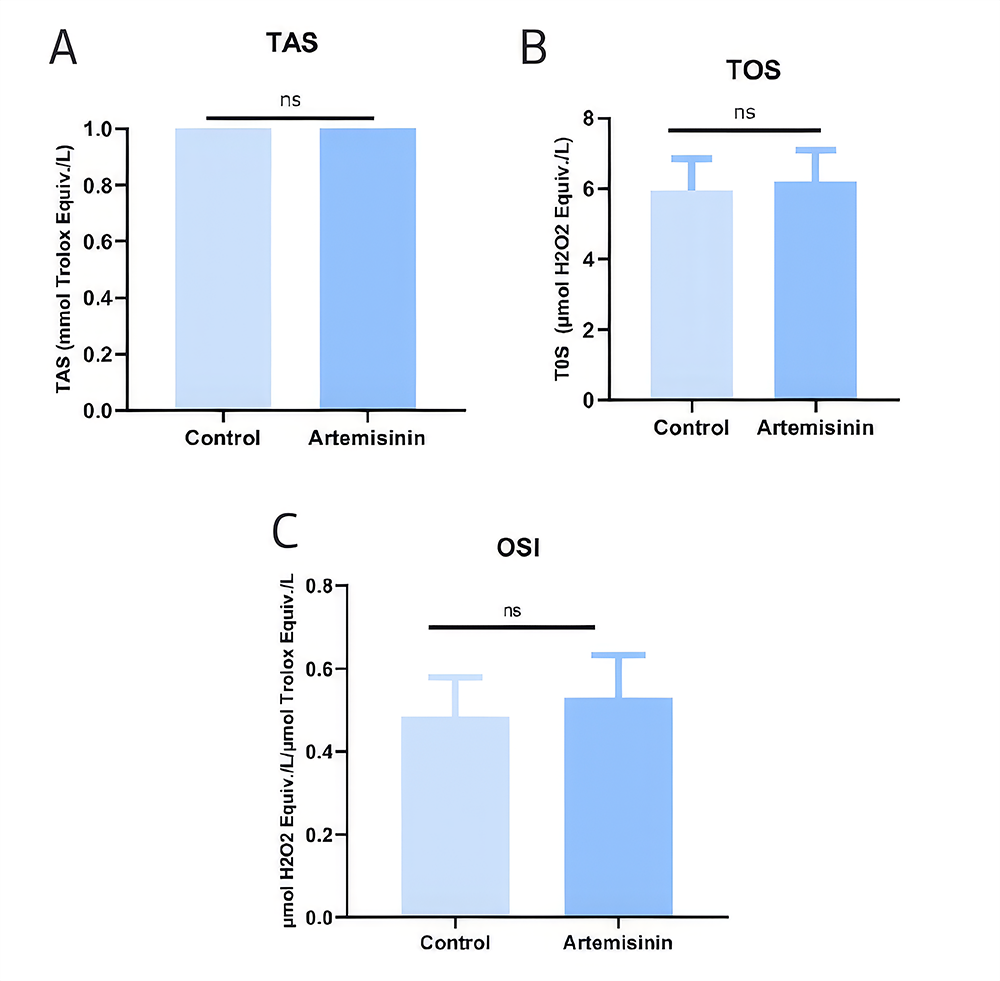
Figure 2. Modulation of the TAS, TOS and OSI values in H1395 lung cancer cells by ART A. TAS (Control group: 1.237 ± 0.059, ART-treated, 1.176 ± 0.056), B. TOS (Control group: 5.949 ± 0.895, ART-treated, 6.203 ± 0.895), C. OSI (Control group: 0.483 ± 0.095, ART-treated: 0.5300± 0.101)
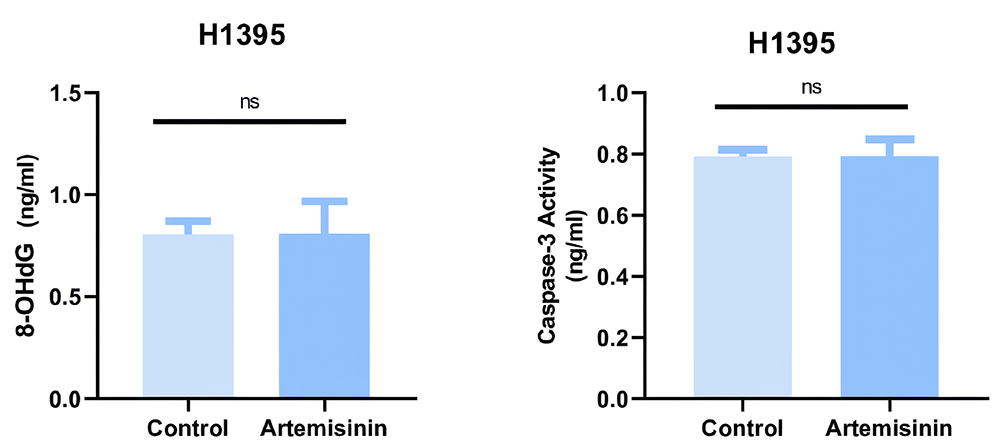
Figure 3. 8-OHdG and Caspase-3 concentration levels of ART-treated H1395 lung cancer cells and controls (8-OHdG Control group 0.805±0.064 ng/mL, ART-treated: 0.809± 0.158 ng/mL), (Caspase-3 Control group: 0.791±0.022, ART-treated 0.793±0.054) (p>0.99)
Tables
Table 1. Real Time PCR forward and reverse primer sequences
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Selen Karaoglanoglu, Gonca Gulbay, Mücahit Seçme. Artemisinin suppresses proliferation and induces oxidative damage and apoptosis in H1395 lung adenocarcinoma cells. Ann Clin Anal Med 2026;17(3):238-243
- Received:
- December 26, 2025
- Accepted:
- February 26, 2026
- Printed:
- March 1, 2026

