Analysis of patients with severe asthma treated with add-on biologic agents
Severe asthma treated with add-on biologic agents
Authors
Abstract
Aim: Severe asthma is a heterogeneous disease associated with frequent exacerbations, impaired lung function, and corticosteroid burden. Although biologic therapies targeting Type 2 inflammation are effective add-on treatments, comparative real-world data remain limited. This study aimed to evaluate the clinical, laboratory, and spirometric effects of biologic therapies in patients with severe asthma.
Methods: This single-center, retrospective observational study included adult patients with severe asthma treated with omalizumab, mepolizumab, or benralizumab between January 2019 and December 2024. Demographic data, Asthma Control Test (ACT) scores, peripheral blood eosinophil count (PBEC), serum total IgE levels, annual exacerbation rates, and spirometric parameters were assessed at baseline and after treatment. Outcomes were analyzed for the overall cohort and by biologic agent.
Results: A total of 178 patients (mean age 40.1 ± 14.0 years; 71.9% female) were included. Biologic therapy significantly improved ACT scores and reduced annual exacerbation rates and PBEC (all p < 0.001). Lung function improved, with a mean increase of approximately 210 mL in FEV₁ and a 9% relative improvement. All biologic agents provided significant clinical benefits, while anti–IL-5 therapies—particularly benralizumab—were associated with greater spirometric improvement compared with anti-IgE therapy.
Conclusion: In this real-world cohort, biologic therapies improved asthma control, reduced exacerbations, and suppressed eosinophilic inflammation in severe asthma, supporting biomarker-driven treatment and suggesting differential lung function effects among biologic agents.
Keywords
Introduction
Asthma is a chronic inflammatory disease of the airways characterized by heterogeneous clinical and biological features and constitutes a substantial burden of morbidity and healthcare expenditure worldwide.1 Although patients whose disease remains uncontrolled despite standard inhaled therapies account for only approximately 5–10% of the overall asthma population, this subgroup is responsible for a disproportionate share of asthma-related complications and healthcare resource utilization.2 According to the Global Initiative for Asthma (GINA) guidelines, severe asthma is defined as asthma that remains uncontrolled despite treatment with high-dose inhaled corticosteroids (ICS) and long-acting β2-agonists (LABA), or that rapidly deteriorates when this treatment is reduced or discontinued.1 Patients with severe asthma experience a markedly increased burden of frequent exacerbations, emergency department visits, and the need for systemic corticosteroids (SCS), along with their associated adverse effects. In particular, long-term use of SCS is associated with serious complications, including osteoporosis, diabetes mellitus, hypertension, increased susceptibility to infections, and cardiovascular disease.3 In recent years, it has become clear that asthma is not a uniform disease but rather a complex syndrome composed of distinct phenotypes and endotypes. Within this framework, a substantial proportion of patients with severe asthma exhibit predominant Type 2 (T2) inflammation, characterized by eosinophilic airway inflammation mediated primarily through interleukin-4 (IL-4), interleukin-5 (IL-5), and interleukin-13 (IL-13) pathways.4,5 Biomarkers such as peripheral blood eosinophil count (PBEC), fractional exhaled nitric oxide (FeNO), and serum total IgE levels reflect the clinical expression of these inflammatory pathways and play a pivotal role in guiding the selection of targeted therapies.6 With the translation of these pathophysiological insights into clinical practice, biologic therapies have ushered in a new era in the management of severe asthma. Biologic agents targeting the anti-IgE, anti–IL-5, anti–IL-5 receptor, and anti–IL-4/IL-13 pathways have been shown to reduce exacerbation frequency, improve lung function and symptom control, and, most importantly, substantially decrease the need for systemic corticosteroids.7,8,9 In addition to evidence from randomized controlled trials, real-world data strongly support the effectiveness and safety of biologic therapies when appropriate patient selection is applied.10,11 Currently, the management of severe asthma has moved away from a “one-size-fits-all” treatment approach toward a personalized, biomarker-driven model. In this context, biologic agents have emerged not only as therapies that improve symptom control but also as targeted treatments with the potential to modify the natural course of the disease.1,12 For optimal management of severe asthma, identifying the right patient, selecting the right biologic agent, and initiating treatment at the right time constitute a critical therapeutic triad.
In our country, three different biologic agents are currently approved for the treatment of severe asthma: omalizumab, mepolizumab, and benralizumab. Among these, omalizumab was the first biologic to receive approval in 2008, followed by mepolizumab in 2019 and benralizumab in 2023. In this study, we aimed to share our clinical experience with patients treated with any of these three biologic agents for severe asthma.
Materials and Methods
Study Design
This study was designed as a single-center, retrospective, observational cohort study evaluating patients who received biologic therapy for severe asthma between January 2019 and December 2024. Adult patients who were diagnosed with severe asthma according to the current GINA criteria and who were treated with at least one biologic agent (omalizumab, mepolizumab, or benralizumab) were included in the study.1
Patients Population
The inclusion criteria for the study were as follows:
(i) age ≥ 18 years;
(ii) a diagnosis of severe asthma that remained uncontrolled despite treatment with high-dose ICS and LABA;
(iii) treatment with a biologic agent for a minimum duration of 12 months; and
(iv) complete availability of clinical, laboratory, and spirometric data.
Patients with chronic pulmonary diseases other than asthma (such as chronic obstructive pulmonary disease or bronchiectasis with predominant symptoms), active malignancy, primary immunodeficiency, eosinophilic granulomatosis with polyangiitis (EGPA), allergic bronchopulmonary aspergillosis (ABPA), asthma–COPD overlap syndrome (ACOS), a previous heavy smoking history (>20 pack-years), those who underwent a biologic treatment switch during follow-up, or those in whom biologic therapy was discontinued shortly after initiation were excluded from the study.
Clinical, Laboratory and Pulmonary Function Assessment
Patients’ demographic characteristics (age and sex), duration of asthma, asthma onset age, presence of concomitant atopic diseases, and exacerbation frequency were retrospectively recorded from medical files. The assessment of atopic status was based on skin prick testing and/or measurement of specific IgE levels, and only patients who were symptomatic underwent evaluation for atopy. Clinical evaluations at baseline and during follow-up were primarily based on Asthma Control Test (ACT) scores. Laboratory assessments included measurements of PBEC and serum total IgE levels. All laboratory analyses were performed in the central laboratory of our institution using standardized methods.
Pulmonary function tests (PFTs) were performed in accordance with the recommendations of the American Thoracic Society (ATS)13 and the European Respiratory Society (ERS). Forced expiratory volume in the first second (FEV₁) was recorded as both absolute values and percentages of predicted values (%). Pre-treatment and follow-up measurements were evaluated comparatively. All laboratory and spirometric data of the patients were recorded using the most recent measurements obtained immediately prior to the initiation of biologic therapy.
Biologic Therapies: Indications and Dosing Regimens
The biologic therapies evaluated in this study included anti-IgE (omalizumab), anti–IL-5 (mepolizumab), and anti–IL-5 receptor alpha (benralizumab). The selection of biologic treatment was based on national reimbursement criteria, GINA recommendations, national asthma guidelines, and the patients’ clinical, functional, and biological characteristics.1,14 The indications for use and dosing regimens of each biologic agent in severe asthma are summarized below.
Omalizumab (anti-IgE)
• Body weight between 20 and 150 kg;
• Persistent uncontrolled symptoms despite treatment with high-dose ICS–LABA and/or leukotriene receptor antagonist (LTRA), along with a history of multiple asthma exacerbations;
• Demonstrated sensitization to at least one perennial allergen—such as house dust mite, cat or dog dander, cockroach, or mold spores—confirmed by a positive skin prick test and/or specific IgE positivity;
• Impaired lung function (FEV₁ < 80% of predicted);
• Serum total IgE levels between 30 and 1500 IU/mL.
• Administered subcutaneously at intervals of 2–4 weeks, with dosing determined according to body weight and serum IgE levels.14
Mepolizumab (anti IL-5)
• PBEC ≥ 300 cells/µL, or ≥ 150 cells/µL in patients receiving long-term maintenance SCS therapy;
• Asthma that remains controlled or uncontrolled despite regular SCS therapy for at least six months and/or uncontrolled asthma despite treatment with a high-dose ICS plus inhaled LABA combination for at least one year, with a history of at least two exacerbations per year requiring systemic corticosteroid use for a minimum of three days
• Administered subcutaneously at a dose of 100 mg every 4 weeks.14
Benralizumab (anti–IL-5 Ra)
• Patients with asthma that remains uncontrolled despite treatment with medium-to-high-dose ICS plus LABA or another controller therapy;
• Patients with eosinophilia (defined as a PBEC ≥ 300 cells/µL, or ≥ 150 cells/µL for patients on regular SCS treatment)
• History of at least two exacerbations in the previous year.
• Administered subcutaneously at a dose of 30 mg, with the first three doses given every 4 weeks, followed by maintenance dosing every 8 weeks.14
Ethical Approval
This study was approved by the Ethics Committee of the University of Health Sciences, Süreyyapaşa Chest Diseases and Thoracic Surgery Training and Research Hospital (Date: 2026-01-20, No: 2024-130).
Statistical Analysis
Statistical analyses were performed using the trial version of SPSS 22.0 (SPSS Inc., Chicago, IL, USA). The normality of distribution for continuous variables was assessed using the Kolmogorov–Smirnov test. Comparisons between independent groups were conducted using the independent samples t-test for normally distributed variables and the Mann–Whitney U test for non-normally distributed variables. For dependent (paired) measurements, the paired samples t-test was applied to variables demonstrating normal distribution, whereas the Wilcoxon signed-rank test was used for variables that did not meet the normality assumption. Associations between categorical variables were evaluated using the chi-square test. Descriptive statistics for normally distributed continuous variables were expressed as mean ± standard deviation, while non-normally distributed continuous variables were presented as median (minimum–maximum). Categorical variables were summarized as frequency and percentage (%). A p-value < 0.05 was considered statistically significant.
Reporting Guidelines
This study was reported in accordance with the STROBE guidelines.
Results
A total of 178 patients with severe asthma receiving biologic therapy were included in the analysis. Treatment distribution comprised omalizumab in 69 patients (39%), mepolizumab in 79 patients (44%), and benralizumab in 30 patients (%17). The cohort had an average age of 40.07 ± 13.99 years, with a clear female predominance (n = 128, 71.9%). Asthma onset occurred at a mean age of 31.41 ± 14.14 years, corresponding to a disease duration of 15.36 ± 10.39 years. The average body mass index was 28.72 ± 6.32 kg/m².
Regarding smoking status, 116 patients were non-smokers, 36 were ex-smokers, and 9 were current smokers. A smoking history of ≥10 pack-years (P/Y)was present in 16.9% of the cohort. Evaluation of comorbidities revealed that allergic rhinitis (AR) was the most frequent comorbidity (63.5%), followed by gastroesophageal reflux disease (GERD) (37.1%), obesity (34.4%), nasal polyposis (NP) (27.5%), and Non-Steroidal Anti-Inflammatory Drug–Exacerbated Respiratory Disease (N-ERD) (10.1%) (Figure 1).
Atopic status was assessed using skin prick testing and/or serum-specific IgE measurements, and 57.3% of patients were identified as atopic. Among atopic patients, house dust mite sensitization was the most common (46.6%), followed by pollen (25.8%), cat and/or dog dander (15.2%), and mold (14.6%) (Figure 2).
Baseline laboratory evaluations showed a median PBEC of 455 cells/µL (min-max: 0–6370) and a median serum total IgE level of 226.50 IU/mL (min-max: 8–3747). In the year prior to initiation of biologic therapy, the mean number of asthma exacerbations requiring at least 3 days of SCS use was 5.99 ± 5.79. The mean ACT score before treatment was 10.47 ± 3.82.
Baseline spirometric assessment demonstrated a mean FEV₁ of 2057.70 ± 880.20 mL (70.32 ± 23.56% predicted). Detailed data are presented in Supplementary Tables 1 and 2.
When pre- and post-treatment values were compared for the entire cohort, a statistically significant reduction was observed in PBEC (median decreased from 210 to 100 cells/µL; p < 0.001) and in the annual number of asthma exacerbations (median decreased from 4 to 0; p < 0.001). In contrast, ACT scores showed a statistically significant improvement (median increased from 10 to 22; p < 0.001). In addition, an approximate increase of 210 mL in FEV₁, corresponding to a relative improvement of 9%, was noted following treatment (Table 1).
When subgroup analyses were performed according to the type of biologic therapy administered, all three biologic agents were associated with statistically significant reductions in PBEC and asthma exacerbation rates, along with a significant improvement in ACT scores. However, comparisons of spirometric parameters revealed differential effects among treatments. Anti–IL-5 therapies (mepolizumab and benralizumab) resulted in statistically significant improvements in FEV₁, both in absolute values (mL) and percentage predicted. In contrast, among patients treated with omalizumab, the increase in FEV₁ expressed in milliliters did not reach statistical significance, whereas a statistically significant improvement was observed in FEV₁ % predicted. Among the three biologic agents, benralizumab was associated with the greatest improvement in FEV₁, both in absolute (mL) and percentage terms. Detailed data are presented in Supplementary Tables 1 and 2.
Discussion
In this study, clinical, functional, and inflammatory responses were comprehensively evaluated in patients with severe asthma receiving biologic therapy. Our findings demonstrate that treatment with omalizumab, mepolizumab, and benralizumab led to significant improvements in asthma control, a marked reduction in exacerbation frequency, and robust suppression of peripheral blood eosinophil levels. These results are consistent with the existing literature, which supports the effectiveness of biologic therapies in the management of severe asthma.1,14
In our study, the improvement in lung function observed in patients treated with benralizumab was particularly noteworthy, as gains in both absolute FEV₁ (mL) and percentage predicted FEV₁ were more pronounced compared with those achieved with other biologic agents. This finding is closely related to the unique mechanism of action of benralizumab. By binding to the IL-5 receptor alpha (IL-5Rα) subunit, benralizumab not only inhibits IL-5–mediated signaling but also induces rapid and near-complete depletion of eosinophils and their precursors through antibody-dependent cell-mediated cytotoxicity.8 This mechanism biologically distinguishes benralizumab from agents such as mepolizumab, which selectively neutralize the IL-5 ligand, and may account for the deeper and more sustained suppression of eosinophilic inflammation observed with benralizumab therapy.15
The potent biological effects of benralizumab have also been clearly demonstrated in the phase 3 randomized controlled SIROCCO and CALIMA trials.8,16 In these studies, benralizumab treatment in patients with severe eosinophilic asthma was associated with significant reductions in annual exacerbation rates and clinically meaningful improvements in FEV₁.8,16 Notably, treatment responses were more pronounced in patients with higher baseline peripheral blood eosinophil counts. In line with these findings, our study showed that PBEC levels in the benralizumab group decreased to near-zero values following treatment, paralleling the marked improvement observed in FEV₁. This observation supports a direct link between effective suppression of eosinophilic inflammation and improvements in airway function.
Previous studies have also reported that therapies targeting the anti–IL-5 pathway may exert more pronounced effects on lung function compared with anti-IgE treatment.17 The primary mechanism of action of omalizumab is based on the suppression of IgE-mediated allergic responses, with a more indirect influence on eosinophilic inflammation.18 Consequently, in patients with a predominantly eosinophilic phenotype, functional gains may remain relatively limited. In our study, although a statistically significant improvement in FEV₁ % predicted was observed in the omalizumab group, the increase in absolute FEV₁ (mL) did not reach statistical significance. This finding may be interpreted as a clinical reflection of the mechanistic differences among biologic agents.
In our study, the significant improvement in ACT scores and the marked reduction in exacerbation frequency observed across all biologic treatment groups indicate that biologic therapies positively influence not only biochemical and functional parameters but also patient-centered clinical outcomes. These findings are consistent with previous studies reporting that biologic agents improve asthma control and have the potential to reduce the need for systemic corticosteroids.19
One of the important contributions of our study to the existing literature is the demonstration that the effectiveness of biologic therapies is maintained beyond highly selected patient populations, extending to patients with severe asthma who exhibit heterogeneous clinical characteristics. Consistent with our findings, previously published observational studies and multicenter analyses have reported that biologic agents yield clinical benefits largely comparable to those observed in randomized controlled trials.20,21 Collectively, these results reinforce the concept that appropriate patient selection based on clinical phenotype and biomarker profiles is a key determinant of treatment response.
Limitations
This study is limited by its single-center, retrospective design and the non-randomized selection of biologic therapies, which may restrict generalizability and introduce potential selection bias.
Conclusion
Biologic therapies significantly improve asthma control, reduce exacerbations, and suppress eosinophilic inflammation in patients with severe asthma. Anti–IL-5 therapies, particularly benralizumab, were associated with greater improvements in lung function, supporting biomarker-driven personalized treatment in real-world practice.
Declarations
Ethics Declarations
This study was approved by the Ethics Committee of the University of Health Sciences, Süreyyapaşa Chest Diseases and Thoracic Surgery Training and Research Hospital (Date: 2026-01-20, No: 130).
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent was waived by the Ethics Committee due to the retrospective design of the study and the use of anonymized patient data.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Author Contributions (CRediT Taxonomy)
Conceptualization: Ş.Ö,F.M.T
Methodology: Ş.Ö,F.M.T
Study Design: Ş.Ö,F.M.T
Data Collection: Ş.Ö,F.M.T
Data Curation: Ş.Ö,F.M.T
Formal Analysis: Ş.Ö,F.M.T
Investigation: Ş.Ö,F.M.T
Resources:Ş.Ö,F.M.T
Writing – Original Draft Preparation: Ş.Ö, F.M.T
Writing – Review & Editing:Ş.Ö,F.M.T
Visualization:Ş.Ö,F.M.T
Supervision: Ş.Ö,F.M.T
Project Administration:Ş.Ö,F.M.T
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
Abbreviations
ABPA: Allergic Bronchopulmonary Aspergillosis
ACOS: Asthma–COPD Overlap Syndrome
ACT: Asthma Control Test
AR: Allergic Rhinitis
ATS: American Thoracic Society
EGPA: Eosinophilic Granulomatosis with Polyangiitis
ERS: European Respiratory Society
FeNO: Fractional Exhaled Nitric Oxide
FEV₁: Forced Expiratory Volume in the First Second
GINA: Global Initiative for Asthma
GERD: Gastroesophageal Reflux Disease
ICS: Inhaled Corticosteroids
IgE: Immunoglobulin E
IL-4: Interleukin-4
IL-5: Interleukin-5
IL-13: Interleukin-13
IL-5Rα: Interleukin-5 Receptor Alpha
LABA: Long-Acting Beta2-Agonists
LTRA: Leukotriene Receptor Antagonist
N-ERD: Non-Steroidal Anti-Inflammatory Drug–Exacerbated Respiratory Disease
PBEC: Peripheral Blood Eosinophil Count
PFT: Pulmonary Function Test
SCS: Systemic Corticosteroids
SPSS: Statistical Package for the Social Sciences
STROBE: Strengthening the Reporting of Observational Studies in Epidemiology
T2: Type 2
References
-
Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention. 2025. Accessed January 19, 2026. https://ginasthma.org
-
Chung KF, Wenzel SE, Brozek JL, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343-373. doi:10.1183/09031936.00202013
-
Dhar R, Rhee CK, Perng DW, et al. The burden of systemic corticosteroid use in asthma management in Asia. Respirology. 2023;28(8):744-757. doi:10.1111/resp.14533
-
Wenzel SE. Asthma phenotypes: the evolution from clinical to molecular approaches. Nat Med. 2012;18(5):716-725. doi:10.1038/nm.2678
-
Fahy JV. Type 2 inflammation in asthma--present in most, absent in many. Nat Rev Immunol. 2015;15(1):57-65. doi:10.1038/nri3786
-
Pavord ID, Holliday M, Reddel HK, et al. Predictive value of blood eosinophils and exhaled nitric oxide in adults with mild asthma: a prespecified subgroup analysis of an open-label, parallel-group, randomised controlled trial. Lancet Respir Med. 2020;8(7):671-680. doi:10.1016/S2213-2600(20)30053-9
-
Busse W, Corren J, Lanier BQ, et al. Omalizumab, anti-IgE recombinant humanized monoclonal antibody, for the treatment of severe allergic asthma. J Allergy Clin Immunol. 2001;108(2):184-190. doi:10.1067/mai.2001.117880
-
Bleecker ER, FitzGerald JM, Chanez P, et al. Efficacy and safety of benralizumab for patients with severe asthma uncontrolled with high-dosage inhaled corticosteroids and long-acting β2-agonists (SIROCCO): a randomised, multicentre, placebo-controlled phase 3 trial. Lancet. 2016;388(10056):2115-2127. doi:10.1016/S0140-6736(16)31324-1
-
Castro M, Corren J, Pavord ID, et al. Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. N Engl J Med. 2018;378(26):2486-2496. doi:10.1056/NEJMoa1804092
-
Canonica GW, Virchow JC, Bourdin A, et al. Real-world comparative effectiveness of biologic therapies in severe asthma: EU-ADVANTAGE. ERJ Open Res. 2025;11(4):01217-2024. doi:10.1183/23120541.01217-2024
-
Perez-de-Llano L, Scelo G, Trung NT, et al. Exploring definitions and predictors of severe asthma clinical remission after biologic treatment in adults. Am J Respir Crit Care Med. 2024;210(7):869-880. doi:10.1164/rccm.202311-2192OC
-
Israel E, Denlinger LC, Bacharier LB, et al. PrecISE: precision medicine in severe asthma: an adaptive platform trial with biomarker ascertainment. J Allergy Clin Immunol. 2021;147(5):1594-1601. doi:10.1016/j.jaci.2021.01.037
-
Graham BL, Steenbruggen I, Miller MR, et al. Standardization of spirometry 2019 update. An official American Thoracic Society and European Respiratory Society technical statement. Am J Respir Crit Care Med. 2019;200(8):e70-e88. doi:10.1164/rccm.201908-1590ST
-
Ulusal Astım Tanı Ve Tedavi Rehberi 2020 Güncellemesi.2020. [National Asthma Diagnosis and Treatment Guideline: 2020 Update.] Accessed January 15, 2026. https://www.aid.org.tr/wp-content/uploads/2020/12/astim-rehberi-2020.pdf
-
Shiomi M, Watanabe R, Ishihara R, et al. Comparative insights on IL-5 targeting with mepolizumab and benralizumab: enhancing EGPA treatment strategies. Biomolecules. 2025;15(4):544. doi:10.3390/biom15040544
-
FitzGerald JM, Bleecker ER, Nair P, et al. Benralizumab, an anti-interleukin-5 receptor α monoclonal antibody, as add-on treatment for patients with severe, uncontrolled, eosinophilic asthma (CALIMA): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2016;388(10056):2128-2141. doi:10.1016/S0140-6736(16)31322-8
-
Pfeffer PE, Ali N, Murray R, et al. Comparative effectiveness of anti-IL5 and anti-IgE biologic classes in patients with severe asthma eligible for both. Allergy. 2023;78(7):1934-1948. doi:10.1111/all.15711
-
Gyawali B, Georas SN, Khurana S. Biologics in severe asthma: a state-of-the-art review. Eur Respir Rev. 2025;34(175):240088. doi:10.1183/16000617.0088-2024
-
Manson SC, Brown RE, Cerulli A, et al. The cumulative burden of oral corticosteroid side effects and the economic implications of steroid use. Respir Med. 2009;103(7):975-994. doi:10.1016/j.rmed.2009.01.003
-
Albers FC, Müllerová H, Gunsoy NB, et al. Biologic treatment eligibility for real-world patients with severe asthma: the IDEAL study. J Asthma. 2018;55(2):152-160. doi:10.1080/02770903.2017.1322611
-
Kavanagh JE, Hearn AP, Dhariwal J, et al. Real-world effectiveness of benralizumab in severe eosinophilic asthma. Chest. 2021;159(2):496-506. doi:10.1016/j.chest.2020.08.2083
Figures
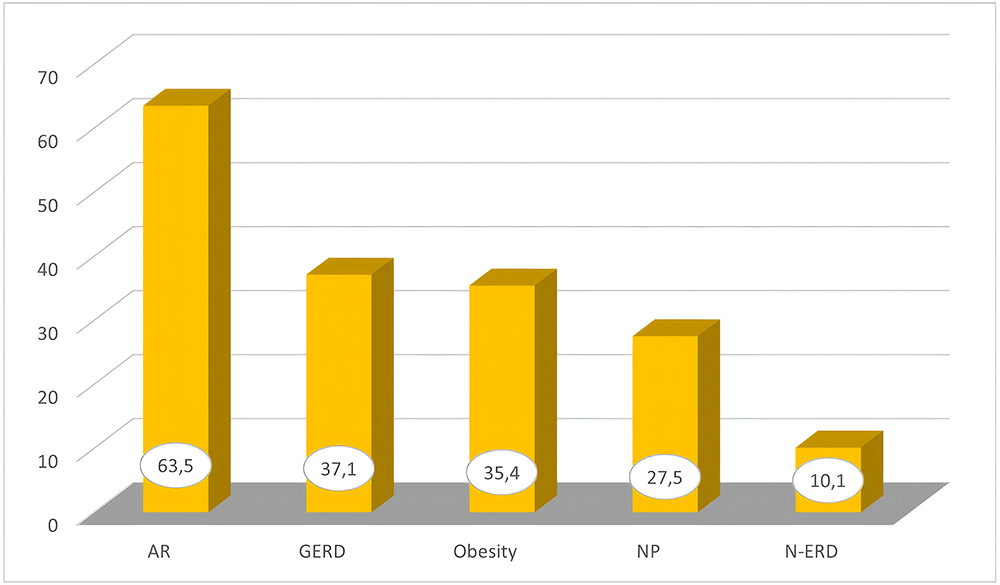
Figure 1. Distribution of comorbidities among the study patients
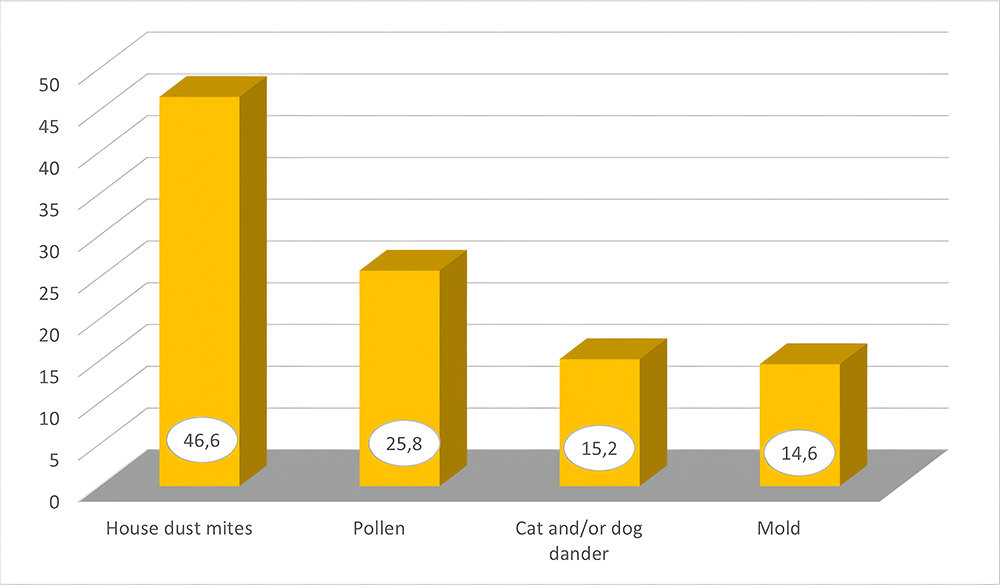
Figure 2. Distribution of sensitizing agents among atopic diseases
Tables
Table 1. Clinical, laboratory, and spirometric changes before and after treatment in all patients
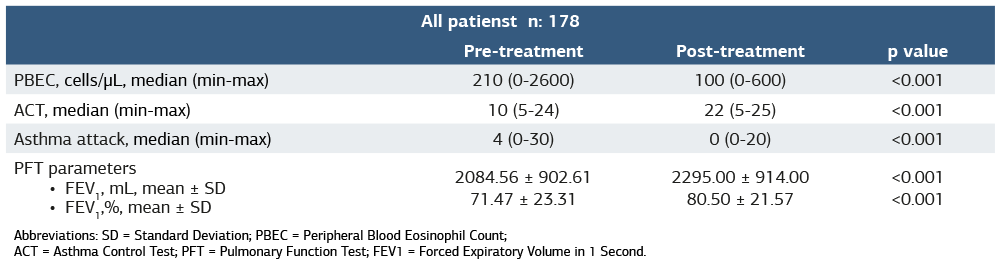
Abbreviations: SD = Standard Deviation; PBEC = Peripheral Blood Eosinophil Count; ACT = Asthma Control Test; PFT = Pulmonary Function Test; FEV1 = Forced Expiratory Volume in 1 Second.
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Şeyma Özden, Fatma Merve Tepetam. Analysis of patients with severe asthma treated with add-on biologic agents. Ann Clin Anal Med 2026; 10.4328/ACAM.50046. Ann Clin Anal Med 2026;17(3):259-264
- Received:
- January 21, 2026
- Accepted:
- February 21, 2026
- Published Online:
- February 28, 2026

