Aortic calcification as a risk factor for pancreatic fistula after distal pancreatectomy
Aortic calcification & POPF after distal pancreatectomy
Authors
Abstract
Aim This study evaluated the association between postoperative pancreatic fistula (POPF) after distal pancreatectomy (DP) and abdominal aortic calcification score (AACS) measured on preoperative computed tomography (CT).
Methods A retrospective review was conducted of patients who underwent DP between 2017 and 2023 in a single tertiary center. POPF was defined according to the 2016 International Study Group of Pancreatic Fistula (ISGPF) criteria. AACS was calculated using the Agatston method, and its relationship with POPF was analyzed alongside established risk factors.
Results Thirty-five patients (57.1% male; mean age 58.4 ± 15.9 years) were included. The median body mass index was 22.9 kg/m², and the mean prognostic nutritional index was 38.5 ± 11.6. Median AACS values above and below the renal artery showed no significant differences between patients with and without POPF (p = 0.658, p = 0.122).
Conclusion No significant correlation was found between AACS and POPF after DP. These findings suggest that AACS is not a reliable predictor of POPF, highlighting the need for novel risk assessment tools to improve postoperative outcomes.
Keywords
Introduction
The International Study Group on Pancreatic Fistula (ISGPF) defines postoperative pancreatic fistula (POPF) as fluid drainage with amylase activity over three times the normal limit after the third postoperative day, affecting patient well-being.1 POPF is common after distal pancreatec-tomy (DP), with an incidence of 5–40%, and can lead to severe outcomes such as bleeding, sepsis, organ failure, longer hospital stays, and death. It remains a significant clinical issue, re-quiring effective prevention and treatment. Risk factors include younger age, low serum albu-min, longer surgery, higher body mass index (BMI), non-pancreatic cancers, soft or thick pancreatic tissue, open surgery, splenectomy, multivisceral resections, and vascular problems.2
Coronary artery disease (CAD) is an independent risk factor for POPF.3 CAD often links with abdominal aortic calcifications, which impair visceral perfusion by altering vascular structure and blood flow, potentially affecting abdominal surgery outcomes. Research shows that a high Abdominal Aortic Calcification Score (AACS) predicts POPF after pancreaticoduodenectomy (PD) in patients over 70.4 We hypothesized that abdominal aortic calcifications could also be a risk factor for POPF after DP across all ages.
The primary outcome of the study was the occurrence of POPF. The secondary outcome was AACS. Accordingly, the primary aim was to investigate the association between elevated abdominal aortic calcification and the development of POPF, while the secondary aim was to evaluate the other factors that may influence the development of POPF in this study population.
Materials and Methods
Clinical Data CollectionData were extracted from the Probel Hospital Information Management System (version 2.1, Izmir, Türkiye) database. Preoperative computed tomography (CT) scans were reviewed using Probel PACS software (Picture Archiving and Communication Systems, version 2.1.65, Izmir, Türkiye), which was connected to the database. Preoperative CT scans were obtained using spiral CT and evaluated via PACS software with a slice thickness of 3 mm.
Study Design• Inclusion Criteria
This retrospective study evaluated patients who underwent DP from 2017 to 2023 at the Health Sciences University, Izmir Tepecik Training and Research Hospital (Figure 1). Exclusion Criteria
Patients without accessible preoperative CT images were excluded from the study.
Classification and EvaluationAccording to the ISGPF 2016 criteria, participants were categorized into two groups based on the presence of POPF. The evaluation included demographic data, general characteristics, radiological features, surgical details, and operative outcomes.
Demographic Data and Patient CharacteristicsThe gathered data comprised age, gender, body mass index (BMI), Prognostic Nutritional Index (PNI), smoking status, and preoperative hemoglobin levels.
Radiological FindingsThe radiological characteristics assessed included the contour of the pancreas (smooth or serrated) and main pancreatic duct caliber on CT, the location of calcific plaques in relation to the renal artery, and the Abdominal Aortic Calcification Score (AACS) for each plaque.
Perioperative FindingsPerioperative data included the type of procedure performed (laparoscopic or open), the method of pancreatic stump closure (suture, stapler, or both), the occurrence of multi-visceral resection, splenectomy, and the total operative time.
Postoperative FindingsPostoperative evaluations assessed hemoglobin levels, the occurrence of hemoglobin re-duction, and pathology outcomes, which were classified as pancreatic tumor, non-pancreatic tumor, or no tumor present. The statistical relationship between AACS and the onset of POPF following DP, in addition to the previously mentioned risk factors, was analyzed and assessed.
Abdominal Aortic Calcification ScoreThe score was calculated manually on PACS using the Agatston method, which involves measuring density in a 1 mm² region of interest (ROI) that exhibits the highest density in each calcific plaque observed on CT, expressed in Hounsfield Units (HU). The scoring system works as follows: 1 point for densities between 130-200 HU, 2 points for densities between 200-300 HU, 3 points for densities between 300-400 HU, and 4 points for densities exceeding 400 HU. The total score is calculated by adding the scores of individual plaques. In this study, calcifications were assessed separately from the abdominal aorta (up to the level of the dia-phragmatic hiatus) to the renal artery and from the renal artery to the aortic-iliac bifurcation.
Ethical ApprovalThis study was approved by the Ethics Committee of Health Sciences University, Izmir Tepecik Training and Research Hospital (Date: 2024-01-10, No: 2023/12-06).
Statistical AnalysisStatistical analyses were conducted using SPSS version 25.0 software. Descriptive statis-tics were reported as mean ± standard deviation for normally distributed variables and median (Q1-Q3) for non-normally distributed variables. Frequencies and percentages were calculated for demographic characteristics. For continuous data, a t-test was performed for normally dis-tributed variables, while the Mann-Whitney U test was used for non-normally distributed varia-bles. Categorical data were analyzed using Pearson’s chi-square or Fisher’s exact tests. Uni-variate analysis was performed to identify the potential risk factors, followed by multivariate analysis (binary logistic regression) to pinpoint independent factors. A p<0.05 was considered statistically significant.
Reporting GuidelinesThis study was conducted and reported in accordance with the STROBE guidelines.
Results
The study included 35 patients with a median age of 58 years, of whom 22.8% developed postoperative pancreatic fistula (POPF), with no significant gender difference. Comparative analysis revealed no statistical differences between groups in terms of body mass index, pancreatic contour, preoperative hemoglobin, tumor type, multivisceral resection with splenectomy, surgical approach, stump closure technique, operative time, smoking status, or perioperative hemoglobin levels. Similarly, no significant association was observed between POPF and ab-dominal aortic calcification scores (AACS) measured above or below the renal artery. Binary logistic regression analysis revealed no independent risk factors associated with complications following distal pancreatectomy. The detailed data are provided in Supplementary Table 1 and Table 2.
Discussion
This study examined how clinical and preoperative factors relate to POPF after distal pan-createctomy. No significant links were found for BMI, pancreatic contour, preoperative hemo-globin, multivisceral resection, or smoking. These findings underscore POPF's complexity and the difficulty in identifying reliable predictors. Although often cited as risk factors, these varia-bles had no measurable effect here, implying other biological or technical factors may be more important.
This study found no significant demographic risk factors. The higher pancreatic fistula rate in some patients may relate to technical difficulty, male body habitus, or older age. Literature offers limited insights into sociodemographic impacts.
Previous studies have suggested that high BMI and comorbidities, reflected by CCI and American Society of Anesthesiologists (ASA) scores, may increase the risk of postoperative complications, including POPF.5 For instance, Khachfe et al.6 reported that obesity negatively affects surgical outcomes in pancreatic surgery, particularly by increasing fistula formation. In contrast, our results did not confirm these associations, which may be explained by differences in patient characteristics, surgical techniques, or the thresholds applied to define high-risk fac-tors.
Pancreatic contour was identified as a potential risk factor for POPF, likely due to its influ-ence on tissue strength and healing. However, evidence remains inconsistent. While some stud-ies, including Maeda et al.,7 suggest an association between soft pancreatic texture and in-creased POPF risk, others report no significant correlation.8,9,10 These discrepancies may reflect differences in surgical techniques, particularly the type of anastomosis performed, such as pancreatico-jejunostomy or pancreatico-gastrostomy.
Although multivisceral resection is often considered a marker of higher surgical risk, our study did not demonstrate a significant association with POPF. This finding aligns with Ramia et al.,11 who similarly reported no correlation between multivisceral resection and POPF inci-dence. The impact of such procedures may instead be influenced by factors such as the extent of resection, underlying pathology, and surgical technique.
Findings showed laparoscopic PD did not increase POPF risk. Review of studies found no significant difference in POPF rates between minimally invasive (laparoscopic and robotic) and open PD.12,13
Regarding splenic preservation in our study, we found no difference in POPF develop-ment. A meta-analysis showed lower POPF rates with spleen-preserved procedures.14 The sur-geons hypothesized that more complicated malignant tumors often require splenectomy, possi-bly increasing POPF risk, which we didn't detect in our single-center study. POPF after DP remains a challenge, with prevention often ineffective. Key factors include the pancreatic stump closure technique and pancreatic anatomy.15 In our study, the closure method wasn't significant-ly linked to POPF occurrence.
Pastena et al. reported preoperative and intraoperative risk scores for POPF after DP, with prolonged operation time as a major risk factor.16 However, our study did not find this, possibly due to the limited sample size and non-homogeneous cases.
Our study found no link between AACS and POPF after DP. Kakizawa et al.17 first meas-ured AACS using the Agatston method in pancreatoduodenectomy patients, finding a signifi-cant link only in those over 70, where 46.9% were elderly, higher than prior studies. Their out-comes for the elderly were similar to those of younger patients. Our patients were generally younger, with only 20% over 70, likely explaining the lower AACS and lack of association. This demographic difference may also have limited the ability to detect a link.
Given the lack of a universal risk scoring system and mixed findings on some risk factors, some may still be unidentified for POPF after DP. Tissue perfusion plays a key role in healing, making preoperative parameters that assess perfusion disorders and POPF risk crucial.18,19 However, this study found no link between tissue perfusion and POPF.
POPF remains an ongoing problem. Identifying risk factors for pancreatic leakage and evaluating preoperative risk factors are key to preventing POPF or reducing morbidity.20 This is the first study linking AACS and POPF in DP patients. Future research should focus on homo-geneous groups and larger samples to develop better strategies.
In our study, we evaluated risk factors previously assessed in meta-analyses, including plaque in the abdominal aorta and AACS. Although literature links age, operation time, high BMI, open surgery, splenectomy, and multi-visceral resection to POPF after DP,2 we found no such relationship. Additionally, calcific plaque in the abdominal aorta above or below the renal artery was not associated with POPF.
Limitations
Its strengths include a focused investigation of the relationship between the AACS and POPF after DP, which is a novel area. The use of the validated Agatston method for AACS adds rigor and reproducibility. The study also includes detailed demographic and perioperative data, offer-ing a comprehensive risk factor analysis. Limitations involve its single-center, retrospective design, which may cause selection bias and limit generalizability. The small sample size (n = 35) reduces statistical power, especially in multivariate analysis. The young cohort and low high AACS ratio might have prevented detecting significant results. Variations in surgical tech-niques, postoperative management, and lack of long-term follow-up could also influence out-comes, but were not fully considered. Future multicenter, prospective studies with larger, di-verse cohorts are needed to validate these findings and identify more risk factors for POPF.
Conclusion
Our findings show that factors like BMI, CCI, ASA, pancreatic contour, hemoglobin levels, multivisceral resection, and smoking, known as risk factors for POPF in past studies, were not significantly linked in this cohort. This underscores the need for more research to find reliable, personalized predictors for POPF after distal pancreatectomy. Advances in understanding POPF's pathophysiology, surgical techniques, and perioperative care may help refine risk fac-tors and enhance outcomes.
Declarations
Ethics Declarations
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study protocol was reviewed and approved by the relevant institutional ethics committee, and all data were handled in compliance with patient confidentiality and data protection principles.
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was waived due to the retrospective design of the study and the use of anonymized patient data.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
None.
Author Contributions (CRediT Taxonomy)
Conceptualization: B.E., G.A.
Methodology: B.E.
Formal Analysis: K.T.
Investigation: F.D.
Data Curation: F.D.
Writing – Original Draft Preparation: B.E.
Writing – Review & Editing: G.A.
Supervision: G.A.
Scientific Responsibility Statement
The authors declare that they are responsible for the article’s scientific content, including study design, data collection, analysis and interpretation, writing, and some of the main line, or all of the preparation and scientific review of the contents, and approval of the final version of the article.
AI Usage Disclosure
No artificial intelligence tools were used in the preparation of this manuscript.
Abbreviations
AACS: Abdominal Aortic Calcification Score
ASA: American Society of Anesthesiologists
BMI: Body Mass Index
CAD: Coronary Artery Disease
CT: Computed Tomography
DP: Distal Pancreatectomy
HU: Hounsfield Unit
ISGPF: International Study Group of Pancreatic Fistula
PACS: Picture Archiving and Communication Systems
PNI: Prognostic Nutritional Index
POPF: Postoperative Pancreatic Fistula
References
-
Bassi C, Marchegiani G, Dervenis C, et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery. 2017;161(3):584-591. doi:10.1016/j.surg.2016.11.014
-
Chong E, Ratnayake B, Lee S, et al. Systematic review and meta-analysis of risk factors of postoperative pancreatic fistula after distal pancreatectomy in the era of 2016 International Study Group pancreatic fistula definition. HPB (Oxford). 2021;23(8):1139-1151. doi:10.1016/j.hpb.2021.02.015
-
Zweig BM, Sheth M, Simpson S, et al. Association of abdominal aortic calcium with coro-nary artery calcium and obstructive coronary artery disease: a pilot study. Int J Cardiovasc Imaging. 2012;28(2):399-404. doi:10.1007/s10554-011-9818-1
-
Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M, Detrano R. Quantifica-tion of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 1990;15(4):827-832. doi:10.1016/0735-1097(90)90282-t
-
Kawaida H, Kono H, Hosomura N, et al. Surgical techniques and postoperative manage-ment to prevent postoperative pancreatic fistula after pancreatic surgery. World J Gastroen-terol. 2019;25(28):3722-3737. doi:10.3748/wjg.v25.i28.3722
-
Khachfe HH, Hammad AY, AlMasri S, et al. Obesity is associated with increased risk for adverse postoperative outcomes after distal pancreatectomy for pancreatic ductal adenocar-cinoma. J Surg Res. 2023;284:164-172. doi:10.1016/j.jss.2022.11.050
-
Maeda K, Kuriyama N, Yuge T, et al. Risk factor analysis of postoperative pancreatic fistu-la after distal pancreatectomy, with a focus on pancreas-visceral fat CT value ratio and ser-rated pancreatic contour. BMC Surg. 2022;22(1):240. doi:10.1186/s12893-022-01650-8
-
Kusafuka T, Kato H, Iizawa Y, et al. Pancreas-visceral fat CT value ratio and serrated panc-reatic contour are strong predictors of postoperative pancreatic fistula after pancreaticojeju-nostomy. BMC Surg. 2020;20(1):129. doi:10.1186/s12893-020-00785-w
-
Tranchart H, Gaujoux S, Rebours V, et al. A preoperative CT scan helps to predict the oc-currence of severe pancreatic fistula after pancreaticoduodenectomy. Ann Surg. 2012;256(1):139-145. doi:10.1097/SLA.0b013e318256c32c
-
Madankan A, Jaliliyan A, Khalili P, et al. Association of preoperative CT-scan features and clinically relevant postoperative pancreatic fistula after pancreaticoduodenectomy: a meta-analysis. ANZ J Surg. 2024;94(6):1030-1038. doi:10.1111/ans.19033
-
Ramia JM, Del Río-Martín JV, Blanco-Fernández G, et al. Distal pancreatectomy with mul-tivisceral resection: a retrospective multicenter study – case series. Int J Surg. 2020;82:123-129. doi:10.1016/j.ijsu.2020.08.024
-
Correa-Gallego C, Dinkelspiel HE, Sulimanoff I, et al. Minimally-invasive vs open pancre-aticoduodenectomy: systematic review and meta-analysis. J Am Coll Surg. 2014;218(1):129-139. doi:10.1016/j.jamcollsurg.2013.09.005
-
Wang S, Shi N, You L, et al. Minimally invasive surgical approach versus open procedure for pancreaticoduodenectomy: a systematic review and meta-analysis. Medicine (Baltimo-re). 2017;96(26):e8619. doi:10.1097/MD.0000000000008619
-
Shi N, Liu SL, Li YT, You L, Dai MH, Zhao YP. Splenic preservation versus splenectomy during distal pancreatectomy: a systematic review and meta-analysis. Ann Surg Oncol. 2016;23(2):365-374. doi:10.1245/s10434-015-4870-z
-
Karabicak I, Satoi S, Yanagimoto H, et al. Comparison of surgical outcomes of three diffe-rent stump closure techniques during distal pancreatectomy. Pancreatology. 2017;17(3):497-503. doi:10.1016/j.pan.2017.04.005
-
De Pastena M, van Bodegraven EA, Mungroop TH, et al. Distal Pancreatectomy Fistula Risk Score (D-FRS): development and international validation. Ann Surg. 2023;277(5):e1099-e1105. doi:10.1097/SLA.0000000000005497
-
Kakizawa N, Noda H, Watanabe F, Ichida K, Suzuki K, Rikiyama T. A high abdominal aortic calcification score on CT is a risk factor for postoperative pancreatic fistula in elderly patients undergoing pancreaticoduodenectomy. World J Surg. 2018;42(4):1129-1137. doi:10.1007/s00268-017-4240-z
-
Turrini O, Paye F, Bachellier P, et al. Pancreatectomy for adenocarcinoma in elderly pati-ents: postoperative outcomes and long-term results: a study of the French Surgical Associa-tion. Eur J Surg Oncol. 2013;39(2):171-178. doi:10.1016/j.ejso.2012.08.017
-
El Nakeeb A, Atef E, El Hanafy E, et al. Outcomes of pancreaticoduodenectomy in elderly patients. Hepatobiliary Pancreat Dis Int. 2016;15(4):419-427. doi:10.1016/s1499-3872(16)60105-4
-
Bonsdorff A, Ghorbani P, Helanterä I, et al. Development and external validation of DIS-PAIR fistula risk score for clinically relevant postoperative pancreatic fistula risk after distal pancreatectomy. Br J Surg. 2022;109(12):1131-1139. doi:10.1093/bjs/znac266
Figures
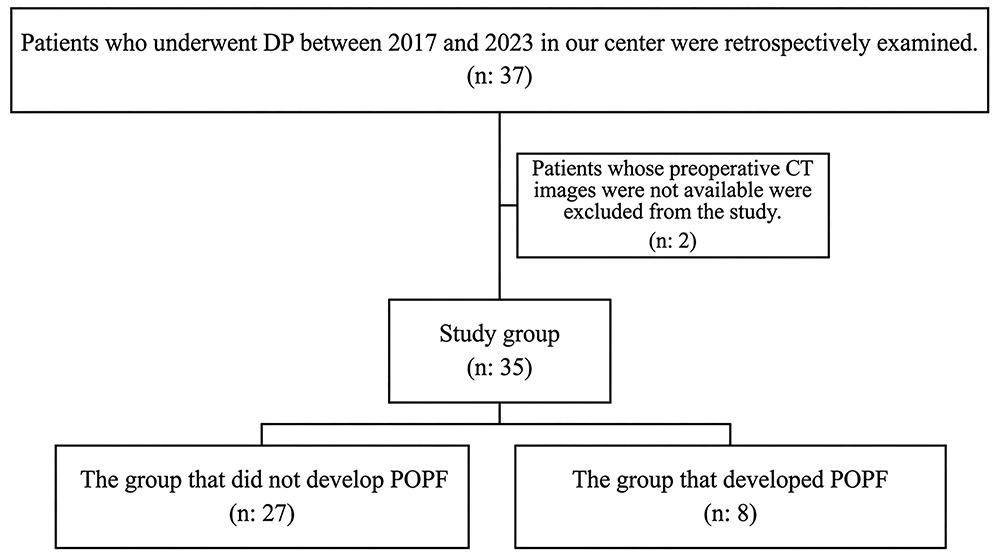
Figure 1. Flow chart of study design
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Goksever Akpinar, Batuhan Eyduran, Korhan Tuncer, Fatma Dikiser. Aortic calcification as a risk factor for pancreatic fistula after distal pancreatectomy. Ann Clin Anal Med 2026;17(Suppl 2):S192-195. doi: 10.4328/ACAM.50101
- Received:
- February 27, 2026
- Accepted:
- March 17, 2026
- Published Online:
- March 17, 2026
- Printed:
- March 20, 2026

